Neural timing is linked to speech perception in noise
- PMID: 20371812
- PMCID: PMC2862599
- DOI: 10.1523/JNEUROSCI.0107-10.2010
Neural timing is linked to speech perception in noise
Abstract
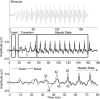
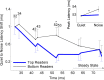
Understanding speech in background noise is challenging for every listener, including those with normal peripheral hearing. This difficulty is attributable in part to the disruptive effects of noise on neural synchrony, resulting in degraded representation of speech at cortical and subcortical levels as reflected by electrophysiological responses. These problems are especially pronounced in clinical populations such as children with learning impairments. Given the established effects of noise on evoked responses, we hypothesized that listening-in-noise problems are associated with degraded processing of timing information at the brainstem level. Participants (66 children; ages, 8-14 years; 22 females) were divided into groups based on their performance on clinical measures of speech-in-noise (SIN) perception and reading. We compared brainstem responses to speech syllables between top and bottom SIN and reading groups in the presence and absence of competing multitalker babble. In the quiet condition, neural response timing was equivalent between groups. In noise, however, the bottom groups exhibited greater neural delays relative to the top groups. Group-specific timing delays occurred exclusively in response to the noise-vulnerable formant transition, not to the more perceptually robust, steady-state portion of the stimulus. These results demonstrate that neural timing is disrupted by background noise and that greater disruptions are associated with the inability to perceive speech in challenging listening conditions.
Figures

References
-
- Akhoun I, Gallégo S, Moulin A, Ménard M, Veuillet E, Berger-Vachon C, Collet L, Thai-Van H. The temporal relationship between speech auditory brainstem responses and the acoustic pattern of the phoneme /ba/ in normal-hearing adults. Clin Neurophysiol. 2008;119:922–933. - PubMed
-
- Benasich AA, Tallal P. Infant discrimination of rapid auditory cues predicts later language impairment. Behav Brain Res. 2002;136:31–49. - PubMed
-
- Bench J, Kowal Ã, Bamford J. The BKB (Bamford-Kowal-Bench) sentence lists for partially-hearing children. Brit J Audiol. 1979;13:108–112. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
