Metronomic chemotherapy enhances antitumor effects of cancer vaccine by depleting regulatory T lymphocytes and inhibiting tumor angiogenesis
- PMID: 20372107
- PMCID: PMC2889744
- DOI: 10.1038/mt.2010.34
Metronomic chemotherapy enhances antitumor effects of cancer vaccine by depleting regulatory T lymphocytes and inhibiting tumor angiogenesis
Abstract
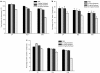
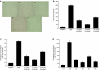
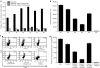
Although cancer vaccines are emerging as innovative methods for cancer treatment, these alone have limited potential for treating measurable tumor burden. Thus, the importance of identifying anticancer strategies with greater potency is necessary. The chimeric DNA vaccine CTGF/E7 (connective tissue growth factor linked to the tumor antigen human papillomavirus 16 E7) generates potent E7-specific immunity and antitumor effects. We tested immune-modulating doses of chemotherapy in combination with the CTGF/E7 DNA vaccine to treat existing tumors in mice. Metronomic low doses of paclitaxel, not the maximal tolerable dose, are synergistic with the antigen-specific DNA vaccine. Paclitaxel, given in metronomic sequence with the CTGF/E7 DNA vaccine enhanced the vaccine's potential to delay tumor growth and decreased metastatic tumors in vivo better than the CTGF/E7 DNA vaccine alone. The two possible mechanisms of metronomic paclitaxel chemotherapy are the depletion of regulatory T cells and the inhibition of tumor angiogenesis rather than direct cancer cell cytolytic effects. Results indicate that combination treatment of metronomic chemotherapy and antigen-specific DNA vaccine can induce more potent antigen-specific immune responses and antitumor effects. This provides an immunologic basis for further testing in cancer patients.
Figures

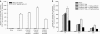
Similar articles
-
Connective tissue growth factor linked to the E7 tumor antigen generates potent antitumor immune responses mediated by an antiapoptotic mechanism.Gene Ther. 2008 Jul;15(13):1007-16. doi: 10.1038/gt.2008.25. Epub 2008 Mar 20. Gene Ther. 2008. PMID: 18356819
-
Cyclophosphamide, doxorubicin, and paclitaxel enhance the antitumor immune response of granulocyte/macrophage-colony stimulating factor-secreting whole-cell vaccines in HER-2/neu tolerized mice.Cancer Res. 2001 May 1;61(9):3689-97. Cancer Res. 2001. PMID: 11325840
-
Immune checkpoint Ab enhances the antigen-specific anti-tumor effects by modulating both dendritic cells and regulatory T lymphocytes.Cancer Lett. 2019 Mar 1;444:20-34. doi: 10.1016/j.canlet.2018.11.039. Epub 2018 Dec 10. Cancer Lett. 2019. PMID: 30543813
-
Metronomic chemotherapy: A potent macerator of cancer by inducing angiogenesis suppression and antitumor immune activation.Cancer Lett. 2017 Aug 1;400:243-251. doi: 10.1016/j.canlet.2016.12.018. Epub 2016 Dec 23. Cancer Lett. 2017. PMID: 28017892 Review.
-
Metronomic chemotherapy and immunotherapy in cancer treatment.Cancer Lett. 2017 Aug 1;400:282-292. doi: 10.1016/j.canlet.2017.01.040. Epub 2017 Feb 9. Cancer Lett. 2017. PMID: 28189534 Review.
Cited by
-
Control of advanced cancer: the road to chronicity.Int J Environ Res Public Health. 2011 Mar;8(3):683-97. doi: 10.3390/ijerph8030683. Epub 2011 Mar 1. Int J Environ Res Public Health. 2011. PMID: 21556173 Free PMC article. Review.
-
Immuno-genomic profiling of biopsy specimens predicts neoadjuvant chemotherapy response in esophageal squamous cell carcinoma.Cell Rep Med. 2022 Aug 16;3(8):100705. doi: 10.1016/j.xcrm.2022.100705. Epub 2022 Aug 8. Cell Rep Med. 2022. PMID: 35944530 Free PMC article.
-
Low-dose paclitaxel improves the therapeutic efficacy of recombinant adenovirus encoding CCL21 chemokine against murine cancer.Cancer Sci. 2014 Nov;105(11):1393-401. doi: 10.1111/cas.12537. Cancer Sci. 2014. PMID: 25230206 Free PMC article.
-
Phase I-II study of everolimus and low-dose oral cyclophosphamide in patients with metastatic renal cell cancer.BMC Cancer. 2011 Nov 30;11:505. doi: 10.1186/1471-2407-11-505. BMC Cancer. 2011. PMID: 22129044 Free PMC article. Clinical Trial.
-
Connective tissue growth factor in tumor pathogenesis.Fibrogenesis Tissue Repair. 2012 Jun 6;5(Suppl 1):S8. doi: 10.1186/1755-1536-5-S1-S8. eCollection 2012. Fibrogenesis Tissue Repair. 2012. PMID: 23259759 Free PMC article.
References
-
- Boon T, Cerottini JC, Van den Eynde B, van der Bruggen P., and , Van Pel A. Tumor antigens recognized by T lymphocytes. Annu Rev Immunol. 1994;12:337–365. - PubMed
-
- Nigam A, Yacavone RF, Zahurak ML, Johns CM, Pardoll DM, Piantadosi S, et al. Immunomodulatory properties of antineoplastic drugs administered in conjunction with GM-CSF-secreting cancer cell vaccines. Int J Oncol. 1998;12:161–170. - PubMed
-
- Kerbel RS, Klement G, Pritchard KI., and , Kamen B. Continuous low-dose anti-angiogenic/ metronomic chemotherapy: from the research laboratory into the oncology clinic. Ann Oncol. 2002;13:12–15. - PubMed
-
- Mekhail TM., and , Markman M. Paclitaxel in cancer therapy. Expert Opin Pharmacother. 2002;3:755–766. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

