Analyses of the specificity of CD4 T cells during the primary immune response to influenza virus reveals dramatic MHC-linked asymmetries in reactivity to individual viral proteins
- PMID: 20373997
- PMCID: PMC2883523
- DOI: 10.1089/vim.2009.0099
Analyses of the specificity of CD4 T cells during the primary immune response to influenza virus reveals dramatic MHC-linked asymmetries in reactivity to individual viral proteins
Abstract
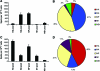
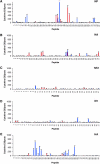
Influenza is a contagious, acute respiratory disease that is a major cause of morbidity and mortality throughout the world. CD4 T cells play an important role in the immune response to this pathogen through the secretion of antiviral cytokines, and by providing help to CD8 T cells and B cells to promote the development of immunological memory and neutralizing antibody responses. Despite these well-defined roles in the anti-influenza response, our understanding of CD4 T-cell diversity and specificity remains limited. In the study reported here, overlapping peptides representing 5 different influenza viral proteins were used in EliSpot assays to enumerate and identify the specificity of anti-influenza CD4 T cells directly ex vivo following infection of mice with influenza virus, using two strains that express unrelated MHC class II molecules. These experiments evaluated whether the reactivity of CD4 T cells generally tracked with particular influenza proteins, or whether MHC preferences were the predominant factor dictating anti-CD4 T-cell specificity in the primary immune response. We made the unexpected discovery that the distribution of CD4 T-cell specificities for different influenza proteins varied significantly depending on the single class II molecule expressed in vivo. In SJL mice, the majority of epitopes were specific for the HA protein, while the NP protein dominated the response in C57BL/10 mice. Given the diversity of human MHC class II molecules, these findings have important implications for the ability to rationally design a vaccine that will generate a specific CD4 T-cell immune response that is effective across diverse human populations.
Figures

Comment in
-
The quality of papers in Viral Immunology continues to rise.Viral Immunol. 2010 Apr;23(2):125-6. doi: 10.1089/vim.2010.Editorial23.2. Viral Immunol. 2010. PMID: 20373992 No abstract available.
Similar articles
-
Loss in CD4 T-cell responses to multiple epitopes in influenza due to expression of one additional MHC class II molecule in the host.Immunology. 2012 Aug;136(4):425-36. doi: 10.1111/j.1365-2567.2012.03599.x. Immunology. 2012. PMID: 22747522 Free PMC article.
-
Identification of HLA class II H5N1 hemagglutinin epitopes following subvirion influenza A (H5N1) vaccination.Vaccine. 2009 Aug 27;27(39):5393-401. doi: 10.1016/j.vaccine.2009.06.081. Epub 2009 Jul 9. Vaccine. 2009. PMID: 19596415
-
Influenza A virus infection-induced macroautophagy facilitates MHC class II-restricted endogenous presentation of an immunodominant viral epitope.FEBS J. 2021 May;288(10):3164-3185. doi: 10.1111/febs.15654. Epub 2020 Dec 18. FEBS J. 2021. PMID: 33830641
-
CD4 T cells in protection from influenza virus: Viral antigen specificity and functional potential.Immunol Rev. 2018 Jul;284(1):91-105. doi: 10.1111/imr.12662. Immunol Rev. 2018. PMID: 29944766 Free PMC article. Review.
-
Memory CD4 T cells in influenza.Curr Top Microbiol Immunol. 2015;386:399-421. doi: 10.1007/82_2014_401. Curr Top Microbiol Immunol. 2015. PMID: 25005927 Free PMC article. Review.
Cited by
-
CD4 T cell help is limiting and selective during the primary B cell response to influenza virus infection.J Virol. 2014 Jan;88(1):314-24. doi: 10.1128/JVI.02077-13. Epub 2013 Oct 23. J Virol. 2014. PMID: 24155379 Free PMC article.
-
T Cell Immunity against Influenza: The Long Way from Animal Models Towards a Real-Life Universal Flu Vaccine.Viruses. 2021 Jan 28;13(2):199. doi: 10.3390/v13020199. Viruses. 2021. PMID: 33525620 Free PMC article. Review.
-
Loss in CD4 T-cell responses to multiple epitopes in influenza due to expression of one additional MHC class II molecule in the host.Immunology. 2012 Aug;136(4):425-36. doi: 10.1111/j.1365-2567.2012.03599.x. Immunology. 2012. PMID: 22747522 Free PMC article.
-
Mutation in Hemagglutinin Antigenic Sites in Influenza A pH1N1 Viruses from 2015-2019 in the United States Mountain West, Europe, and the Northern Hemisphere.Genes (Basel). 2022 May 19;13(5):909. doi: 10.3390/genes13050909. Genes (Basel). 2022. PMID: 35627294 Free PMC article.
-
Detection of site-specific positive Darwinian selection on pandemic influenza A/H1N1 virus genome: integrative approaches.Genetica. 2013 Jun;141(4-6):143-55. doi: 10.1007/s10709-013-9713-x. Epub 2013 Mar 26. Genetica. 2013. PMID: 23529677
References
-
- World Health Organization. New WHO influenza vaccine recommendations for 2005–2006 season, northern hemisphere. Euro Surveill. 2005;10:E050217–E050211. - PubMed
-
- American Academy of Pediatrics. Prevention of influenza: recommendations for influenza immunization of children, 2007–2008. Pediatrics. 2008;121:e1016–e1031. - PubMed
-
- Centers for Disease Control and Prevention. Influenza vaccination coverage among children and adults—United States, 2008–2009 influenza season. MMWR Morb Mortal Wkly Rep. 2009;58:1091–1095. - PubMed
-
- Centers for Disease Control and Prevention. Serum cross-reactive antibody response to a novel influenza A (H1N1) virus after vaccination with seasonal influenza vaccine. MMWR Morb Mortal Wkly Rep. 2009;58:521–524. - PubMed
-
- Anthony DD. Lehmann PV. T-cell epitope mapping using the ELISPOT approach. Methods. 2003;29:260–269. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

