Adhesion to and biofilm formation on IB3-1 bronchial cells by Stenotrophomonas maltophilia isolates from cystic fibrosis patients
- PMID: 20374629
- PMCID: PMC2858031
- DOI: 10.1186/1471-2180-10-102
Adhesion to and biofilm formation on IB3-1 bronchial cells by Stenotrophomonas maltophilia isolates from cystic fibrosis patients
Abstract
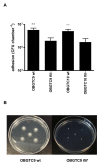
Background: Stenotrophomonas maltophilia has recently gained considerable attention as an important emerging pathogen in cystic fibrosis (CF) patients. However, the role of this microorganism in the pathophysiology of CF lung disease remains largely unexplored. In the present study for the first time we assessed the ability of S. maltophilia CF isolates to adhere to and form biofilm in experimental infection experiments using the CF-derived bronchial epithelial IB3-1cell line. The role of flagella on the adhesiveness of S. maltophilia to IB3-1 cell monolayers was also assessed by using fliI mutant derivative strains.
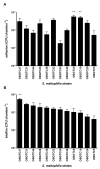
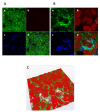
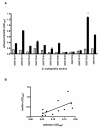
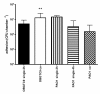
Results: All S. maltophilia CF isolates tested in the present study were able, although at different levels, to adhere to and form biofilm on IB3-1 cell monolayers. Scanning electron and confocal microscopy revealed S. maltophilia structures typical of biofilm formation on bronchial IB3-1 cells. The loss of flagella significantly (P < 0.001) decreased bacterial adhesiveness, if compared to that of their parental flagellated strains. S. maltophilia CF isolates were also able to invade IB3-1 cells, albeit at a very low level (internalization rate ranged from 0.01 to 4.94%). Pre-exposure of IB3-1 cells to P. aeruginosa PAO1 significantly increased S. maltophilia adhesiveness. Further, the presence of S. maltophilia negatively influenced P. aeruginosa PAO1 adhesiveness.
Conclusions: The main contribution of the present study is the finding that S. maltophilia is able to form biofilm on and invade CF-derived IB3-1 bronchial epithelial cells, thus posing a rationale for the persistence and the systemic spread of this opportunistic pathogen in CF patients. Experiments using in vivo models which more closely mimic CF pulmonary tissues will certainly be needed to validate the relevance of our results.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

