Enzymatic hydrolysis of trehalose dimycolate releases free mycolic acids during mycobacterial growth in biofilms
- PMID: 20375425
- PMCID: PMC2878501
- DOI: 10.1074/jbc.M110.112813
Enzymatic hydrolysis of trehalose dimycolate releases free mycolic acids during mycobacterial growth in biofilms
Abstract
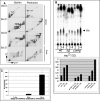
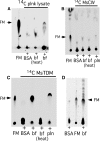
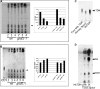
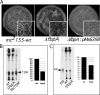
Mycobacterial species, like other microbes, spontaneously form multicellular drug-tolerant biofilms when grown in vitro in detergent-free liquid media. The structure of Mycobacterium tuberculosis biofilms is formed through genetically programmed pathways and is built upon a large abundance of novel extracellular free mycolic acids (FM), although the mechanism of FM synthesis remained unclear. Here we show that the FM in Mycobacterium smegmatis biofilms is produced through the enzymatic release from constitutively present mycolyl derivatives. One of the precursors for FM is newly synthesized trehalose dimycolate (TDM), which is cleaved by a novel TDM-specific serine esterase, Msmeg_1529. Disruption of Msmeg_1529 leads to undetectable hydrolytic activity, reduced levels of FM in the mutant, and retarded biofilm growth. Furthermore, enzymatic hydrolysis of TDM remains conserved in M. tuberculosis, suggesting the presence of a TDM-specific esterase in this pathogen. Overall, this study provides the first evidence for an enzymatic release of free mycolic acids from cell envelope mycolates during mycobacterial growth.
Figures

Similar articles
-
Exposure to a cutinase-like serine esterase triggers rapid lysis of multiple mycobacterial species.J Biol Chem. 2013 Jan 4;288(1):382-92. doi: 10.1074/jbc.M112.419754. Epub 2012 Nov 15. J Biol Chem. 2013. PMID: 23155047 Free PMC article.
-
Granuloma formation activity and mycolic acid composition of mycobacterial cord factor.Cell Mol Life Sci. 1997 Mar;53(3):227-32. doi: 10.1007/pl00000595. Cell Mol Life Sci. 1997. PMID: 9104485 Free PMC article.
-
Mycobacteriophage Ms6 LysB specifically targets the outer membrane of Mycobacterium smegmatis.Microbiology (Reading). 2010 May;156(Pt 5):1497-1504. doi: 10.1099/mic.0.032821-0. Epub 2010 Jan 21. Microbiology (Reading). 2010. PMID: 20093291 Free PMC article.
-
Pathway to synthesis and processing of mycolic acids in Mycobacterium tuberculosis.Clin Microbiol Rev. 2005 Jan;18(1):81-101. doi: 10.1128/CMR.18.1.81-101.2005. Clin Microbiol Rev. 2005. PMID: 15653820 Free PMC article. Review.
-
Towards understanding the functional diversity of cell wall mycolic acids of Mycobacterium tuberculosis.Prog Lipid Res. 2012 Oct;51(4):325-39. doi: 10.1016/j.plipres.2012.05.002. Epub 2012 May 30. Prog Lipid Res. 2012. PMID: 22659327 Review.
Cited by
-
Comparison of Ultrastructure, Extracellular Matrix, and Drug Susceptibility in M. avium subs. hominissuis Biofilms.Pathogens. 2023 Dec 8;12(12):1427. doi: 10.3390/pathogens12121427. Pathogens. 2023. PMID: 38133310 Free PMC article.
-
Molecular Mechanisms of MmpL3 Function and Inhibition.Microb Drug Resist. 2023 May;29(5):190-212. doi: 10.1089/mdr.2021.0424. Epub 2023 Feb 21. Microb Drug Resist. 2023. PMID: 36809064 Free PMC article. Review.
-
A hydrolase of trehalose dimycolate induces nutrient influx and stress sensitivity to balance intracellular growth of Mycobacterium tuberculosis.Cell Host Microbe. 2014 Feb 12;15(2):153-63. doi: 10.1016/j.chom.2014.01.008. Cell Host Microbe. 2014. PMID: 24528862 Free PMC article.
-
The anaplerotic node is essential for the intracellular survival of Mycobacterium tuberculosis.J Biol Chem. 2018 Apr 13;293(15):5695-5704. doi: 10.1074/jbc.RA118.001839. Epub 2018 Feb 23. J Biol Chem. 2018. PMID: 29475946 Free PMC article.
-
Antimycobacterial Precatorin A Flavonoid Displays Antibiofilm Activity against Mycobacterium bovis BCG.ACS Omega. 2023 Oct 20;8(43):40665-40676. doi: 10.1021/acsomega.3c05703. eCollection 2023 Oct 31. ACS Omega. 2023. PMID: 37929145 Free PMC article.
References
-
- Hall-Stoodley L., Stoodley P. (2002) Curr. Opin. Biotechnol. 13, 228–233 - PubMed
-
- Costerton J. W., Irvin R. T., Cheng K. J. (1981) Crit. Rev. Microbiol. 8, 303–338 - PubMed
-
- Blankenship J. R., Mitchell A. P. (2006) Curr. Opin. Microbiol. 9, 588–594 - PubMed
-
- Kolter R., Greenberg E. P. (2006) Nature 441, 300–302 - PubMed
-
- Kolter R., Losick R. (1998) Science 280, 226–227 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

