Endoplasmic reticulum stress and atherosclerosis
- PMID: 20376052
- PMCID: PMC2897068
- DOI: 10.1038/nm0410-396
Endoplasmic reticulum stress and atherosclerosis
Abstract
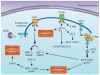
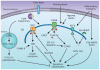
Atherosclerosis and related cardiovascular diseases represent one of the greatest threats to human health worldwide. Despite important progress in prevention and treatment, these conditions still account for one third of all deaths annually. Often presented together with obesity, insulin resistance and type 2 diabetes, these chronic diseases are strongly influenced by pathways that lie at the interface of chronic inflammation and nutrient metabolism. Here I discuss recent advances in the study of endoplasmic reticulum stress as one mechanism that links immune response with nutrient sensing in the pathogenesis of atherosclerosis and its complications.
Figures
Similar articles
-
The role of endoplasmic reticulum stress in the progression of atherosclerosis.Circ Res. 2010 Oct 1;107(7):839-50. doi: 10.1161/CIRCRESAHA.110.224766. Circ Res. 2010. PMID: 20884885 Free PMC article. Review.
-
Mechanisms of ER stress-induced apoptosis in atherosclerosis.Arterioscler Thromb Vasc Biol. 2011 Dec;31(12):2792-7. doi: 10.1161/ATVBAHA.111.224881. Arterioscler Thromb Vasc Biol. 2011. PMID: 22096099 Free PMC article. Review.
-
Obesity: stressing about unfolded proteins.Nat Med. 2010 Apr;16(4):374-6. doi: 10.1038/nm0410-374. Nat Med. 2010. PMID: 20376042 No abstract available.
-
Endoplasmic reticulum stress and the inflammatory basis of metabolic disease.Cell. 2010 Mar 19;140(6):900-17. doi: 10.1016/j.cell.2010.02.034. Cell. 2010. PMID: 20303879 Free PMC article. Review.
-
Methods and models for monitoring UPR-associated macrophage death during advanced atherosclerosis.Methods Enzymol. 2011;489:277-96. doi: 10.1016/B978-0-12-385116-1.00016-9. Methods Enzymol. 2011. PMID: 21266236 Free PMC article.
Cited by
-
Effect of endoplasmic reticulum (ER) stress inducer thapsigargin on the expression of extracellular-superoxide dismutase in mouse 3T3-L1 adipocytes.J Clin Biochem Nutr. 2013 Mar;52(2):101-5. doi: 10.3164/jcbn.12-46. Epub 2013 Feb 1. J Clin Biochem Nutr. 2013. PMID: 23525536 Free PMC article.
-
Anti-inflammatory strategies for plaque stabilization after acute coronary syndromes.Curr Atheroscler Rep. 2013 Jun;15(6):327. doi: 10.1007/s11883-013-0327-7. Curr Atheroscler Rep. 2013. PMID: 23636864 Review.
-
"Inflammaging" as a Druggable Target: A Senescence-Associated Secretory Phenotype-Centered View of Type 2 Diabetes.Oxid Med Cell Longev. 2016;2016:1810327. doi: 10.1155/2016/1810327. Epub 2016 Jun 1. Oxid Med Cell Longev. 2016. PMID: 27340505 Free PMC article. Review.
-
Foam cell-derived 4-hydroxynonenal induces endothelial cell senescence in a TXNIP-dependent manner.J Cell Mol Med. 2015 Aug;19(8):1887-99. doi: 10.1111/jcmm.12561. Epub 2015 Mar 5. J Cell Mol Med. 2015. PMID: 25754218 Free PMC article.
-
Vitamin D suppression of endoplasmic reticulum stress promotes an antiatherogenic monocyte/macrophage phenotype in type 2 diabetic patients.J Biol Chem. 2012 Nov 9;287(46):38482-94. doi: 10.1074/jbc.M112.386912. Epub 2012 Sep 24. J Biol Chem. 2012. PMID: 23012375 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

