A human-specific de novo protein-coding gene associated with human brain functions
- PMID: 20376170
- PMCID: PMC2845654
- DOI: 10.1371/journal.pcbi.1000734
A human-specific de novo protein-coding gene associated with human brain functions
Abstract
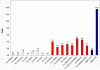
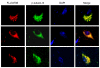
To understand whether any human-specific new genes may be associated with human brain functions, we computationally screened the genetic vulnerable factors identified through Genome-Wide Association Studies and linkage analyses of nicotine addiction and found one human-specific de novo protein-coding gene, FLJ33706 (alternative gene symbol C20orf203). Cross-species analysis revealed interesting evolutionary paths of how this gene had originated from noncoding DNA sequences: insertion of repeat elements especially Alu contributed to the formation of the first coding exon and six standard splice junctions on the branch leading to humans and chimpanzees, and two subsequent substitutions in the human lineage escaped two stop codons and created an open reading frame of 194 amino acids. We experimentally verified FLJ33706's mRNA and protein expression in the brain. Real-Time PCR in multiple tissues demonstrated that FLJ33706 was most abundantly expressed in brain. Human polymorphism data suggested that FLJ33706 encodes a protein under purifying selection. A specifically designed antibody detected its protein expression across human cortex, cerebellum and midbrain. Immunohistochemistry study in normal human brain cortex revealed the localization of FLJ33706 protein in neurons. Elevated expressions of FLJ33706 were detected in Alzheimer's brain samples, suggesting the role of this novel gene in human-specific pathogenesis of Alzheimer's disease. FLJ33706 provided the strongest evidence so far that human-specific de novo genes can have protein-coding potential and differential protein expression, and be involved in human brain functions.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




Similar articles
-
[Analysis, identification and correction of some errors of model refseqs appeared in NCBI Human Gene Database by in silico cloning and experimental verification of novel human genes].Yi Chuan Xue Bao. 2004 May;31(5):431-43. Yi Chuan Xue Bao. 2004. PMID: 15478601 Chinese.
-
MECP2, a gene associated with Rett syndrome in humans, shows conserved coding regions, independent Alu insertions, and a novel transcript across primate evolution.BMC Genet. 2015 Jul 7;16:77. doi: 10.1186/s12863-015-0240-x. BMC Genet. 2015. PMID: 26148505 Free PMC article.
-
Alport syndrome. Molecular genetic aspects.Dan Med Bull. 2009 Aug;56(3):105-52. Dan Med Bull. 2009. PMID: 19728970
-
MicroRNAs and their therapeutic potential for human diseases: aberrant microRNA expression in Alzheimer's disease brains.J Pharmacol Sci. 2010;114(3):269-75. doi: 10.1254/jphs.10r11fm. Epub 2010 Oct 9. J Pharmacol Sci. 2010. PMID: 20953120 Review.
-
The repeat region of microtubule-associated protein tau forms part of the core of the paired helical filament of Alzheimer's disease.Ann Med. 1989;21(2):127-32. doi: 10.3109/07853898909149199. Ann Med. 1989. PMID: 2504257 Review.
Cited by
-
Turnover of ribosome-associated transcripts from de novo ORFs produces gene-like characteristics available for de novo gene emergence in wild yeast populations.Genome Res. 2019 Jun;29(6):932-943. doi: 10.1101/gr.239822.118. Epub 2019 May 31. Genome Res. 2019. PMID: 31152050 Free PMC article.
-
The HIV-1 Antisense Gene ASP: The New Kid on the Block.Vaccines (Basel). 2021 May 17;9(5):513. doi: 10.3390/vaccines9050513. Vaccines (Basel). 2021. PMID: 34067514 Free PMC article. Review.
-
Evolutionary interrogation of human biology in well-annotated genomic framework of rhesus macaque.Mol Biol Evol. 2014 May;31(5):1309-24. doi: 10.1093/molbev/msu084. Epub 2014 Feb 27. Mol Biol Evol. 2014. PMID: 24577841 Free PMC article.
-
Identification of novel GDNF isoforms and cis-antisense GDNFOS gene and their regulation in human middle temporal gyrus of Alzheimer disease.J Biol Chem. 2011 Dec 30;286(52):45093-102. doi: 10.1074/jbc.M111.310250. Epub 2011 Nov 11. J Biol Chem. 2011. PMID: 22081608 Free PMC article.
-
Evaluating Phylostratigraphic Evidence for Widespread De Novo Gene Birth in Genome Evolution.Mol Biol Evol. 2016 May;33(5):1245-56. doi: 10.1093/molbev/msw008. Epub 2016 Jan 11. Mol Biol Evol. 2016. PMID: 26758516 Free PMC article.
References
-
- Ohno S. Berlin: Springer-Verlag; 1970. Evolutionby gene duplication.
-
- Leister D. Tandem and segmental gene duplication and recombination in the evolution of plant disease resistance gene. Trends Genet. 2004;20:116–122. - PubMed
-
- Long M, Langley CH. Natural selection and the origin of jingwei, a chimeric processed functional gene in Drosophila. Science. 1993;260:91–95. - PubMed
-
- Wang W, Zhang J, Alvarez C, Llopart A, Long M. The origin of the Jingwei gene and the complex modular structure of its parental gene, yellow emperor, in Drosophila melanogaster. Mol Biol Evol. 2000;17:1294–1301. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

