Biochemical and functional properties of distinct nicotinic acetylcholine receptors in the superior cervical ganglion of mice with targeted deletions of nAChR subunit genes
- PMID: 20377613
- PMCID: PMC2989642
- DOI: 10.1111/j.1460-9568.2010.07133.x
Biochemical and functional properties of distinct nicotinic acetylcholine receptors in the superior cervical ganglion of mice with targeted deletions of nAChR subunit genes
Abstract
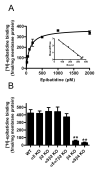
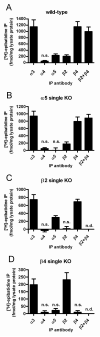
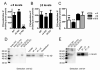
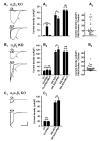
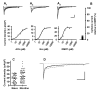
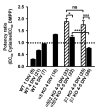
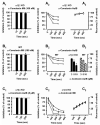
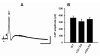
Nicotinic acetylcholine receptors (nAChRs) mediate fast synaptic transmission in ganglia of the autonomic nervous system. Here, we determined the subunit composition of hetero-pentameric nAChRs in the mouse superior cervical ganglion (SCG), the function of distinct receptors (obtained by deletions of nAChR subunit genes) and mechanisms at the level of nAChRs that might compensate for the loss of subunits. As shown by immunoprecipitation and Western blots, wild-type (WT) mice expressed: alpha 3 beta 4 (55%), alpha 3 beta 4 alpha 5 (24%) and alpha 3 beta 4 beta 2 (21%) nAChRs. nAChRs in beta 4 knockout (KO) mice were reduced to < 15% of controls and no longer contained the alpha 5 subunit. Compound action potentials, recorded from the postganglionic (internal carotid) nerve and induced by preganglionic nerve stimulation, did not differ between alpha 5 beta 4 KO and WT mice, suggesting that the reduced number of receptors in the KO mice did not impair transganglionic transmission. Deletions of alpha 5 or beta2 did not affect the overall number of receptors and we found no evidence that the two subunits substitute for each other. In addition, dual KOs allowed us to study the functional properties of distinct alpha 3 beta4 and alpha 3 beta 2 receptors that have previously only been investigated in heterologous expression systems. The two receptors strikingly differed in the decay of macroscopic currents, the efficacy of cytisine, and their responses to the alpha-conotoxins AuIB and MII. Our data, based on biochemical and functional experiments and several mouse KO models, clarify and significantly extend previous observations on the function of nAChRs in heterologous systems and the SCG.
Figures

References
-
- Alkadhi KA, Alzoubi KH, Aleisa AM. Plasticity of synaptic transmission in autonomic ganglia. Prog. Neurobiol. 2005a;75:83–108. - PubMed
-
- Alkadhi KA, Alzoubi KH, Aleisa AM, Tanner FL, Nimer AS. Psychosocial stress-induced hypertension results from in vivo expression of long-term potentiation in rat sympathetic ganglia. Neurobiol. Dis. 2005b;20:849–857. - PubMed
-
- Brejc K, van Dijk WJ, Schuurmans M, van der Oost J, Smit AG, Sixma TK. Crystal structure of an ACh-binding protein reveals the ligand-binding domain of nicotinic receptors. Nature. 2001;411:269–276. - PubMed
-
- Cartier GE, Yoshikami D, Gray WR, Luo S, Olivera BM, McIntosh JM. A new α-conotoxin which targets α3β2 nicotinic acetylcholine receptors. J. Biol. Chem. 1996;271:7522–7528. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

