Insufficient luteinizing hormone-induced intracellular signaling disrupts ovulation in preovulatory follicles lacking estrogen receptor-{beta}
- PMID: 20378682
- PMCID: PMC2875826
- DOI: 10.1210/en.2009-1446
Insufficient luteinizing hormone-induced intracellular signaling disrupts ovulation in preovulatory follicles lacking estrogen receptor-{beta}
Abstract
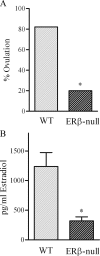
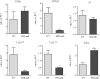
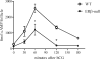
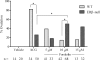
Gonadotropin-stimulated estrogen receptor-beta (ERbeta)-null preovulatory follicles exhibit submaximal estradiol production, insufficient acquisition of LH receptor, and attenuated expression of essential ovulatory genes. These observations lead to low ovulatory rates compared with wild-type (WT) follicles. We hypothesize that insufficient LH receptor results in reduced cAMP production after an ovulatory stimulus. Individual preantral follicles were cultured with FSH for 4 d and then induced to ovulate with a single dose of human chorionic gonadotropin (hCG). cAMP levels 1 h after hCG were 50% lower in ERbeta-null than WT follicles. To determine whether the lack of LH receptor, and resulting lack of cAMP, could be bypassed by direct activation of adenylyl cyclase, WT and ERbeta-null follicles were induced to ovulate with forskolin. Ten micromolar forskolin doubled the ovulatory rate of ERbeta-null follicles compared with treatment with hCG ( approximately 50 vs. 25%, respectively). In WT follicles, 10 microm forskolin reduced the ovulation rate compared with hCG (14 vs. 83%, respectively), indicating that high doses of forskolin inhibited WT ovulation. A 10 microm concentration of forskolin induced cAMP levels in ERbeta-null follicles that were comparable to levels produced in WT follicles after hCG and either partially or completely rescued the attenuated expression of LH-responsive genes. These data indicate that direct activation of adenylyl cyclase, resulting in increased production of cAMP, partially rescues the ovulatory response of ERbeta-null follicles, suggesting that insufficient LH receptor and low cAMP levels contribute to their poor ovulatory rates. We also determined that ERbeta-null ovaries exhibit an alteration in the activation of ERK1/2. Our evaluation of the ERbeta-null ovarian phenotype indicates that ERbeta plays a role in facilitating folliculogenesis. We show that expression of ERbeta in preovulatory follicles is required for adequate cAMP production and propose that an optimal level of cAMP is required for hCG-stimulated ovulation.
Figures



References
-
- Richards JS, Fitzpatrick SL, Clemens JW, Morris JK, Alliston T, Sirois J 1995 Ovarian cell differentiation: a cascade of multiple hormones, cellular signals, and regulated genes. Recent Prog Horm Res 50:223–254 - PubMed
-
- Knecht M, Brodie AM, Catt KJ 1985 Aromatase inhibitors prevent granulosa cell differentiation: an obligatory role for estrogens in luteinizing hormone receptor expression. Endocrinology 117:1156– 1161 - PubMed
-
- Kessel B, Liu YX, Jia XC, Hsueh AJ 1985 Autocrine role of estrogens in the augmentation of luteinizing hormone receptor formation in cultured rat granulosa cells. Biol Reprod 32:1038–1050 - PubMed
-
- Segaloff DL, Wang HY, Richards JS 1990 Hormonal regulation of luteinizing hormone/chorionic gonadotropin receptor mRNA in rat ovarian cells during follicular development and luteinization. Mol Endocrinol 4:1856–1865 - PubMed
-
- Couse JF, Yates MM, Deroo BJ, Korach KS 2005 Estrogen receptor-β is critical to granulosa cell differentiation and the ovulatory response to gonadotropins. Endocrinology 146:3247–3262 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

