Involvement of SLC17A9-dependent vesicular exocytosis in the mechanism of ATP release during T cell activation
- PMID: 20382737
- PMCID: PMC2878504
- DOI: 10.1074/jbc.M110.112417
Involvement of SLC17A9-dependent vesicular exocytosis in the mechanism of ATP release during T cell activation
Abstract
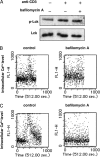
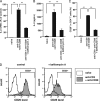
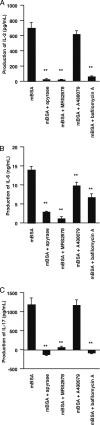
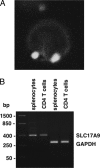
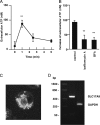
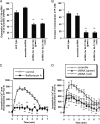
Recent reports have shown that T cell receptor (TCR)-dependent ATP release from T cells is involved in production of interleukin-2 (IL-2) through activation of P2 receptors. Stimulation of TCR induces ATP release from T cells through gap junction hemichannels and maxianion channels, at least in part. However, the mechanisms of ATP release from activated T cells are not fully understood. Here, we studied the mechanisms of ATP release during TCR-dependent T cell activation by investigating the effects of various inhibitors on TCR-dependent ATP release from murine T cells. We found that not only anion channel and gap junction hemichannel inhibitors, but also exocytosis inhibitors suppressed the ATP release. These results suggest that ATP release from murine T cells is regulated by various mechanisms, including exocytosis. An inhibitor of exocytosis, bafilomycin A, significantly blocked TCR signaling, such as Ca(2+) elevation and IL-2 production. Furthermore, bafilomycin A, ectonucleotidase, and P2Y(6) receptor antagonist significantly inhibited production of pro-inflammatory cytokines from external antigen-restimulated splenocytes, indicating that vesicular exocytosis-mediated purinergic signaling has a significant role in TCR-dependent cytokine production. We also detected vesicular ATP in murine T cells and human T lymphoma Jurkat cells, both of which also expressed mRNA of SLC17A9, a vesicular nucleotide transporter. Knockdown of SLC17A9 in Jurkat cells markedly reduced ATP release and cytosolic Ca(2+) elevation after TCR stimulation, suggesting involvement of SLC17A9-dependent vesicular exocytosis in ATP release and T cell activation. In conclusion, vesicular exocytosis of ATP appears to play a role in T cell activation and immune responses.
Figures
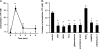

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

