Cardiomyocyte contractile status is associated with differences in fibronectin and integrin interactions
- PMID: 20382852
- PMCID: PMC2886644
- DOI: 10.1152/ajpheart.01156.2009
Cardiomyocyte contractile status is associated with differences in fibronectin and integrin interactions
Abstract
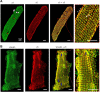
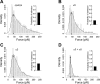
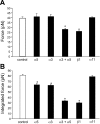
Integrins link the extracellular matrix (ECM) with the intracellular cytoskeleton and other cell adhesion-associated signaling proteins to function as mechanotransducers. However, direct quantitative measurements of the cardiomyocyte mechanical state and its relationship to the interactions between specific ECM proteins and integrins are lacking. The purpose of this study was to characterize the interactions between the ECM protein fibronectin (FN) and integrins in cardiomyocytes and to test the hypothesis that these interactions would vary during contraction and relaxation states in cardiomyocytes. Using atomic force microscopy, we quantified the unbinding force (adhesion force) and adhesion probability between integrins and FN and correlated these measurements with the contractile state as indexed by cell stiffness on freshly isolated mouse cardiomyocytes. Experiments were performed in normal physiological (control), high-K(+) (tonically contracted), or low-Ca(2+) (fully relaxed) solutions. Under control conditions, the initial peak of adhesion force between FN and myocyte alpha(3)beta(1)- and/or alpha(5)beta(1)-integrins was 39.6 +/- 1.3 pN. The binding specificity between FN and alpha(3)beta(1)- and alpha(5)beta(1)-integrins was verified by using monoclonal antibodies against alpha(3)-, alpha(5)-, alpha(3) + alpha(5)-, or beta(1)-integrin subunits, which inhibited binding by 48%, 65%, 70%, or 75%, respectively. Cytochalasin D or 2,3-butanedione monoxime (BDM), to disrupt the actin cytoskeleton or block myofilament function, respectively, significantly decreased the cell stiffness; however, the adhesion force and binding probability were not altered. Tonic contraction with high-K(+) solution increased total cell adhesion (1.2-fold) and cell stiffness (27.5-fold) compared with fully relaxed cells with low-Ca(2+) solution. However, it could be partially prevented by high-K(+) bath solution containing BDM, which suppresses contraction by inhibiting the actin-myosin interactions. Thus, our results demonstrate that integrin binding to FN is modulated by the contractile state of cardiac myocytes.
Figures


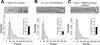
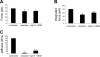

References
-
- Ahumada GG, Saffitz JE. Fibronectin in rat heart: a link between cardiac myocytes and collagen. J Histochem Cytochem 32: 383–388, 1984 - PubMed
-
- Belke DD, Dillmann WH. Altered cardiac calcium handling in diabetes. Curr Hypertens Rep 6: 424–429, 2004 - PubMed
-
- Benoit M, Gabriel D, Gerisch G, Gaub HE. Discrete interactions in cell adhesion measured by single-molecule force spectroscopy. Nat Cell Biol 2: 313–317, 2000 - PubMed
-
- Brancaccio M, Hirsch E, Notte A, Selvetella G, Lembo G, Tarone G. Integrin signalling: the tug-of-war in heart hypertrophy. Cardiovasc Res 70: 422–433, 2006 - PubMed
-
- Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes 54: 1615–1625, 2005 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

