The zinc-finger protein MAZR is part of the transcription factor network that controls the CD4 versus CD8 lineage fate of double-positive thymocytes
- PMID: 20383150
- PMCID: PMC3365445
- DOI: 10.1038/ni.1860
The zinc-finger protein MAZR is part of the transcription factor network that controls the CD4 versus CD8 lineage fate of double-positive thymocytes
Abstract
The CD4 versus CD8 lineage specification of thymocytes is linked to coreceptor expression. The transcription factor MAZR has been identified as an important regulator of Cd8 expression. Here we show that variegated CD8 expression by loss of Cd8 enhancers was reverted in MAZR-deficient mice, which confirms that MAZR negatively regulates the Cd8 loci during the transition to the double-positive (DP) stage. Moreover, loss of MAZR led to partial redirection of major histocompatibility complex (MHC) class I-restricted thymocytes into CD4(+) helper-like T cells, which correlated with derepression of Th-POK, a central transcription factor for helper-lineage development. MAZR bound the silencer of the gene encoding Th-POK, which indicated direct regulation of this locus by MAZR. Thus, MAZR is part of the transcription factor network that regulates the CD8 lineage differentiation of DP thymocytes.
Figures
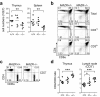
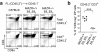
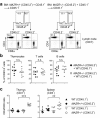
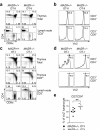



Comment in
-
Another zinc finger in the pie of CD4-CD8 lineage choice.Nat Immunol. 2010 May;11(5):370-1. doi: 10.1038/ni0510-370. Nat Immunol. 2010. PMID: 20404849 No abstract available.
References
-
- Bosselut R. CD4/CD8-lineage differentiation in the thymus: from nuclear effectors to membrane signals. Nat Rev Immunol. 2004;4:529–540. - PubMed
-
- He X, Kappes DJ. CD4/CD8 lineage commitment: light at the end of the tunnel? Curr Opin Immunol. 2006;18:135–142. - PubMed
-
- Ellmeier W, Sawada S, Littman DR. The regulation of CD4 and CD8 coreceptor gene expression during T cell development. Annu Rev Immunol. 1999;17:523–554. - PubMed
-
- Kioussis D, Ellmeier W. Chromatin and CD4, CD8A and CD8B gene expression during thymic differentiation. Nat Rev Immunol. 2002;2:909–919. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

