Simultaneous expression of different transgenes in neurons and glia by combining in utero electroporation with the Tol2 transposon-mediated gene transfer system
- PMID: 20384787
- PMCID: PMC2948423
- DOI: 10.1111/j.1365-2443.2010.01397.x
Simultaneous expression of different transgenes in neurons and glia by combining in utero electroporation with the Tol2 transposon-mediated gene transfer system
Abstract
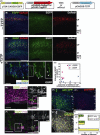
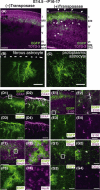
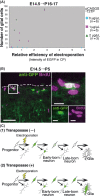
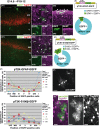
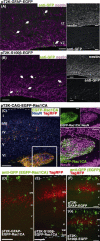
In utero electroporation is widely used to study neuronal development and function by introducing plasmid DNA into neural progenitors during embryogenesis. This is an effective and convenient method of introducing plasmid DNA into neural precursors and is suitable for manipulating gene expression in cells of the CNS. However, the applicability of this technique is comparatively limited to neuronal research, as the plasmid DNA introduced into neural progenitors during embryogenesis is diluted by cell proliferation and is not stably maintained in glial cells generated around and after birth. To overcome this limitation, we applied the Tol2 transposon system, which integrates a transgene into the genome of the host cell, to in utero electroporation. With this system, we confirmed that the transgene was effectively maintained in the progeny of embryonic neural precursors, astrocytes and oligodendrocytes. Using the glial promoters GFAP and S100beta, targeted and stable expressions of transgenes in glia were obtained, which enabled the expression of different transgenes simultaneously in neurons and glia. Glia-targeted expression of the transgene that causes neuronal migration defect was achieved without the defect. Thus, use of the Tol2 transposon system in combination with in utero electroporation is a powerful method for studying glia-neuron interactions in vivo.
Figures
Similar articles
-
Stable integration and conditional expression of electroporated transgenes in chicken embryos.Dev Biol. 2007 May 15;305(2):616-24. doi: 10.1016/j.ydbio.2007.01.043. Epub 2007 Feb 7. Dev Biol. 2007. PMID: 17362912
-
Successful elimination of non-neural cells and unachievable elimination of glial cells by means of commonly used cell culture manipulations during differentiation of GFAP and SOX2 positive neural progenitors (NHA) to neuronal cells.BMC Biotechnol. 2008 Jul 19;8:56. doi: 10.1186/1472-6750-8-56. BMC Biotechnol. 2008. PMID: 18638414 Free PMC article.
-
A method for stable transgenesis of radial glia lineage in rat neocortex by piggyBac mediated transposition.J Neurosci Methods. 2012 Jun 15;207(2):172-80. doi: 10.1016/j.jneumeth.2012.03.016. Epub 2012 Apr 11. J Neurosci Methods. 2012. PMID: 22521325 Free PMC article.
-
Gain- and loss-of-function approaches in the chick embryo.Methods Cell Biol. 2008;87:237-56. doi: 10.1016/S0091-679X(08)00212-4. Methods Cell Biol. 2008. PMID: 18485300 Review.
-
Neurons from radial glia: the consequences of asymmetric inheritance.Curr Opin Neurobiol. 2003 Feb;13(1):34-41. doi: 10.1016/s0959-4388(03)00013-8. Curr Opin Neurobiol. 2003. PMID: 12593980 Review.
Cited by
-
Identification of genes supporting cold resistance of mammalian cells: lessons from a hibernator.Cell Death Dis. 2024 Sep 19;15(9):685. doi: 10.1038/s41419-024-07059-w. Cell Death Dis. 2024. PMID: 39300059 Free PMC article.
-
Differential potentials of neural progenitors for the generation of neurons and non-neuronal cells in the developing amniote brain.Sci Rep. 2019 Mar 14;9(1):4514. doi: 10.1038/s41598-019-40599-2. Sci Rep. 2019. PMID: 30872629 Free PMC article.
-
A tightly controlled conditional knockdown system using the Tol2 transposon-mediated technique.PLoS One. 2012;7(3):e33380. doi: 10.1371/journal.pone.0033380. Epub 2012 Mar 13. PLoS One. 2012. PMID: 22428039 Free PMC article.
-
Recent molecular approaches to understanding astrocyte function in vivo.Front Cell Neurosci. 2013 Dec 24;7:272. doi: 10.3389/fncel.2013.00272. Front Cell Neurosci. 2013. PMID: 24399932 Free PMC article. Review.
-
Association of astrocytes with neurons and astrocytes derived from distinct progenitor domains in the subpallium.Sci Rep. 2015 Jul 20;5:12258. doi: 10.1038/srep12258. Sci Rep. 2015. PMID: 26193445 Free PMC article.
References
-
- Fukuchi-Shimogori T, Grove EA. Neocortex patterning by the secreted signaling molecule FGF8. Science. 2001;294:1071–1074. - PubMed
-
- Funahashi J, Okafuji T, Ohuchi H, Noji S, Tanaka H, Nakamura H. Role of Pax-5 in the regulation of a mid-hindbrain organizer’s activity. Dev Growth Differ. 1999;41:59–72. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

