Translational considerations for cancer nanomedicine
- PMID: 20385183
- PMCID: PMC2921639
- DOI: 10.1016/j.jconrel.2010.04.008
Translational considerations for cancer nanomedicine
Abstract
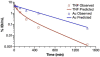
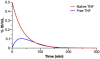
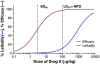
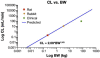
There are many important considerations during preclinical development of cancer nanomedicines, including: 1) unique aspects of animal study design; 2) the difficulties in evaluating biological potency, especially for complex formulations; 3) the importance of analytical methods that can determine platform stability in vivo, and differentiate bound and free active pharmaceutical ingredient (API) in biological matrices; and 4) the appropriateness of current dose scaling techniques for estimation of clinical first-in-man dose from preclinical data. Biologics share many commonalities with nanotechnology products with regard to complexity and biological attributes, and can, in some cases, provide context for dealing with these preclinical issues. In other instances, such as the case of in vivo stability analysis, new approaches are required. This paper will discuss the significance of these preclinical issues, and present examples of current methods and best practices for addressing them. Where possible, these recommendations are justified using the existing regulatory guidance literature.
Copyright 2010 Elsevier B.V. All rights reserved.
Figures


Similar articles
-
Cancer nanomedicines: closing the translational gap.Lancet. 2014 Dec 20;384(9961):2175-6. doi: 10.1016/S0140-6736(14)61457-4. Epub 2014 Dec 19. Lancet. 2014. PMID: 25625382 Free PMC article. No abstract available.
-
Dendrimers in the context of nanomedicine.Int J Pharm. 2020 Jan 5;573:118814. doi: 10.1016/j.ijpharm.2019.118814. Epub 2019 Nov 20. Int J Pharm. 2020. PMID: 31759101 Review.
-
Hyperthermia approaches for enhanced delivery of nanomedicines to solid tumors.Biotechnol Bioeng. 2015 Oct;112(10):1967-83. doi: 10.1002/bit.25653. Epub 2015 Jul 14. Biotechnol Bioeng. 2015. PMID: 25995079 Review.
-
Nanotechnology-based intelligent drug design for cancer metastasis treatment.Biotechnol Adv. 2014 Jul-Aug;32(4):761-77. doi: 10.1016/j.biotechadv.2013.10.013. Epub 2013 Nov 6. Biotechnol Adv. 2014. PMID: 24211475 Review.
-
Can nanomedicines kill cancer stem cells?Adv Drug Deliv Rev. 2013 Nov;65(13-14):1763-83. doi: 10.1016/j.addr.2013.09.016. Epub 2013 Oct 10. Adv Drug Deliv Rev. 2013. PMID: 24120657 Free PMC article. Review.
Cited by
-
Common pitfalls in nanotechnology: lessons learned from NCI's Nanotechnology Characterization Laboratory.Integr Biol (Camb). 2013 Jan;5(1):66-73. doi: 10.1039/c2ib20117h. Integr Biol (Camb). 2013. PMID: 22772974 Free PMC article.
-
Polymeric curcumin nanoparticle pharmacokinetics and metabolism in bile duct cannulated rats.Mol Pharm. 2013 May 6;10(5):1977-87. doi: 10.1021/mp4000019. Epub 2013 Apr 22. Mol Pharm. 2013. PMID: 23534919 Free PMC article.
-
Stable isotope method to measure drug release from nanomedicines.J Control Release. 2015 Dec 28;220(Pt A):169-174. doi: 10.1016/j.jconrel.2015.10.042. Epub 2015 Oct 24. J Control Release. 2015. PMID: 26596375 Free PMC article.
-
Application of a Scavenger Receptor A1-Targeted Polymeric Prodrug Platform for Lymphatic Drug Delivery in HIV.Mol Pharm. 2020 Oct 5;17(10):3794-3812. doi: 10.1021/acs.molpharmaceut.0c00562. Epub 2020 Sep 9. Mol Pharm. 2020. PMID: 32841040 Free PMC article.
-
Triggering RNAi with multifunctional RNA nanoparticles and their delivery.DNA RNA Nanotechnol. 2015 Jan;2(1):1-12. doi: 10.1515/rnan-2015-0001. Epub 2015 Jul 27. DNA RNA Nanotechnol. 2015. PMID: 34322586 Free PMC article.
References
-
- McNeil SE. Nanotechnology for the biologist. J. Leukoc. Biol. 2005;78:585–594. - PubMed
-
- Sandanaraj BS, Gremlich HU, Kneuer R, Dawson J, Wacha S. Fluorescent nanoprobes as a biomarker for increased vascular permeability: implications in diagnosis and treatment of cancer and inflammation. Bioconjugate Chem. 2010;21:93–101. - PubMed
-
- Akiyama Y, Mori T, Katayama Y, Niidome T. The effects of PEG grafting level and injection dose on gold nanorod biodistribution in tumor-bearing mice. J. Control. Release. 2009;139:81–84. - PubMed
-
- Thomas TP, Ye JY, Chang YC, Kotlyar A, Cao Z, Majoros IJ, Norris TB, Baker JR. Investigation of tumor cell targeting of a dendrimer nanoparticle using a double-clad optical fiber probe. J. Biomed. Opt. 2008;13:014024. - PubMed
-
- Hussain S, Pluckthun A, Allen TM, Zangemeister-Wittke U. Antitumor activity of an epithelial cell adhesion molecule targeted nanovesicular drug delivery system. Mol. Cancer Ther. 2007;6:3019–3027. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

