A pooled analysis of the durability and predictors of treatment response of cilostazol in patients with intermittent claudication
- PMID: 20385711
- PMCID: PMC2883185
- DOI: 10.1177/1358863X10361545
A pooled analysis of the durability and predictors of treatment response of cilostazol in patients with intermittent claudication
Abstract
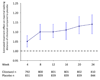
Pharmacologic therapy for intermittent claudication in patients with peripheral artery disease (PAD) is limited. We aimed to determine the durability of cilostazol treatment response over time, treatment effects in various subpopulations, and long-term safety. This analysis pooled original data from nine randomized, controlled trials evaluating cilostazol in intermittent claudication, including 1258 subjects treated with cilostazol 100 mg bid. Analysis of covariance was used to compare differences in walking distance, and a pooled random-effects weighted mean difference in maximal walking distance (MWD) was determined. Temporal effects were analyzed by compiling data at 4-week intervals in studies of 24 weeks in duration. Cilostazol was associated with a 50.7% improvement from baseline in MWD compared with placebo (24.3%), with an absolute improvement of 42.1 meters greater than the improvement with placebo (p < 0.001) over a mean follow-up period of 20.4 weeks. Continued increases were demonstrated over the 24-week treatment period. These benefits were seen in all subgroups, after stratifying by age, sex, smoking status, duration of PAD, diabetes, hypertension, prior myocardial infarction, or beta-blocker use. Cilostazol did not increase the risk of all-cause mortality (RR 0.95 [0.68-1.35]). In conclusion, treatment with cilostazol achieves benefits in walking distance that are sustained at 24 weeks and observed irrespective of baseline clinical characteristics. Cilostazol demonstrated no increased risk of all-cause mortality.
Figures


Similar articles
-
Cilostazol for intermittent claudication.Cochrane Database Syst Rev. 2021 Jun 30;6(6):CD003748. doi: 10.1002/14651858.CD003748.pub5. Cochrane Database Syst Rev. 2021. PMID: 34192807 Free PMC article.
-
Effect of cilostazol on treadmill walking, community-based walking ability, and health-related quality of life in patients with intermittent claudication due to peripheral arterial disease: meta-analysis of six randomized controlled trials.J Am Geriatr Soc. 2002 Dec;50(12):1939-46. doi: 10.1046/j.1532-5415.2002.50604.x. J Am Geriatr Soc. 2002. PMID: 12473004
-
A new pharmacological treatment for intermittent claudication: results of a randomized, multicenter trial.Arch Intern Med. 1999 Sep 27;159(17):2041-50. doi: 10.1001/archinte.159.17.2041. Arch Intern Med. 1999. PMID: 10510990 Clinical Trial.
-
A comparison of cilostazol and pentoxifylline for treating intermittent claudication.Am J Med. 2000 Nov;109(7):523-30. doi: 10.1016/s0002-9343(00)00569-6. Am J Med. 2000. PMID: 11063952 Clinical Trial.
-
Treatment of intermittent claudication: cilostazol.Hosp Med. 2004 Aug;65(8):466-70. doi: 10.12968/hosp.2004.65.8.15491. Hosp Med. 2004. PMID: 15330347 Review.
Cited by
-
Dual Antiplatelet Therapy: A Concise Review for Clinicians.Life (Basel). 2023 Jul 18;13(7):1580. doi: 10.3390/life13071580. Life (Basel). 2023. PMID: 37511955 Free PMC article. Review.
-
Therapeutic angiogenesis using stem cell-derived extracellular vesicles: an emerging approach for treatment of ischemic diseases.Stem Cell Res Ther. 2019 Jun 3;10(1):158. doi: 10.1186/s13287-019-1276-z. Stem Cell Res Ther. 2019. PMID: 31159859 Free PMC article. Review.
-
The Future of Vascular Biology and Medicine.Circulation. 2016 Jun 21;133(25):2603-9. doi: 10.1161/CIRCULATIONAHA.116.023513. Circulation. 2016. PMID: 27324355 Free PMC article. Review. No abstract available.
-
Intermittent claudication: new targets for drug development.Drugs. 2013 Jul;73(10):999-1014. doi: 10.1007/s40265-013-0078-3. Drugs. 2013. PMID: 23775528 Review.
-
Diabetic vasculopathy: macro and microvascular injury.Curr Pathobiol Rep. 2020 Mar;8(1):1-14. doi: 10.1007/s40139-020-00205-x. Epub 2020 Jan 27. Curr Pathobiol Rep. 2020. PMID: 32655983 Free PMC article.
References
-
- Tanaka T, Ishikawa T, Hagiwara M, Onoda K, Itoh H, Hidaka H. Effects of cilostazol, a selective cAMP phosphodiesterase inhibitor on the contraction of vascular smooth muscle. Pharmacology. 1988;36(5):313–320. - PubMed
-
- Oida K, Ebata K, Kanehara H, Suzuki J, Miyamori I. Effect of cilostazol on impaired vasodilatory response of the brachial artery to ischemia in smokers. J Atheroscler Thromb. 2003;10(2):93–98. - PubMed
-
- Igawa T, Tani T, Chijiwa T, Shiragiku T, Shimidzu S, Kawamura K, et al. Potentiation of anti-platelet aggregating activity of cilostazol with vascular endothelial cells. Thromb Res. 1990 Feb 15;57(4):617–623. - PubMed
-
- Woo SK, Kang WK, Kwon KI. Pharmacokinetic and pharmacodynamic modeling of the antiplatelet and cardiovascular effects of cilostazol in healthy humans. Clin Pharmacol Ther. 2002 Apr;71(4):246–252. - PubMed
-
- Beebe HG, Dawson DL, Cutler BS, Herd JA, Strandness DE, Jr, Bortey EB, et al. A new pharmacological treatment for intermittent claudication: results of a randomized, multicenter trial. Arch Intern Med. 1999 Sep 27;159(17):2041–2050. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

