Adrenergic modulation of focal adhesion kinase protects human ovarian cancer cells from anoikis
- PMID: 20389021
- PMCID: PMC2860925
- DOI: 10.1172/JCI40802
Adrenergic modulation of focal adhesion kinase protects human ovarian cancer cells from anoikis
Abstract
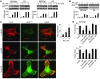
Chronic stress is associated with hormonal changes that are known to affect multiple systems, including the immune and endocrine systems, but the effects of stress on cancer growth and progression are not fully understood. Here, we demonstrate that human ovarian cancer cells exposed to either norepinephrine or epinephrine exhibit lower levels of anoikis, the process by which cells enter apoptosis when separated from ECM and neighboring cells. In an orthotopic mouse model of human ovarian cancer, restraint stress and the associated increases in norepinephrine and epinephrine protected the tumor cells from anoikis and promoted their growth by activating focal adhesion kinase (FAK). These effects involved phosphorylation of FAKY397, which was itself associated with actin-dependent Src interaction with membrane-associated FAK. Importantly, in human ovarian cancer patients, behavioral states related to greater adrenergic activity were associated with higher levels of pFAKY397, which was in turn linked to substantially accelerated mortality. These data suggest that FAK modulation by stress hormones, especially norepinephrine and epinephrine, can contribute to tumor progression in patients with ovarian cancer and may point to potential new therapeutic targets for cancer management.
Figures
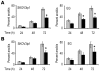

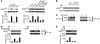


References
-
- Grippo AJ, Moffitt JA, Johnson AK. Cardiovascular alterations and autonomic imbalance in an experimental model of depression. Am J Physiol Regul Integr Comp Physiol. 2002;282(5):R1333–R1341. - PubMed
-
- Riley V. The role of stress in malignancy. Cancer Detect Prev. 1979;2:235–255.
Publication types
MeSH terms
Substances
Grants and funding
- T32CA101642/CA/NCI NIH HHS/United States
- F31CA126474/CA/NCI NIH HHS/United States
- R01 CA109298/CA/NCI NIH HHS/United States
- R01 CA110793/CA/NCI NIH HHS/United States
- CA110793/CA/NCI NIH HHS/United States
- CA-104825/CA/NCI NIH HHS/United States
- R01 CA104825/CA/NCI NIH HHS/United States
- A152737/PHS HHS/United States
- F31 CA126474/CA/NCI NIH HHS/United States
- P50 CA083639/CA/NCI NIH HHS/United States
- R01 CA140933/CA/NCI NIH HHS/United States
- T32 CA101642/CA/NCI NIH HHS/United States
- CA109298/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

