Heterosubtypic neutralizing antibodies are produced by individuals immunized with a seasonal influenza vaccine
- PMID: 20389023
- PMCID: PMC2860935
- DOI: 10.1172/JCI41902
Heterosubtypic neutralizing antibodies are produced by individuals immunized with a seasonal influenza vaccine
Abstract
The target of neutralizing antibodies that protect against influenza virus infection is the viral protein HA. Genetic and antigenic variation in HA has been used to classify influenza viruses into subtypes (H1-H16). The neutralizing antibody response to influenza virus is thought to be specific for a few antigenically related isolates within a given subtype. However, while heterosubtypic antibodies capable of neutralizing multiple influenza virus subtypes have been recently isolated from phage display libraries, it is not known whether such antibodies are produced in the course of an immune response to influenza virus infection or vaccine. Here we report that, following vaccination with seasonal influenza vaccine containing H1 and H3 influenza virus subtypes, some individuals produce antibodies that cross-react with H5 HA. By immortalizing IgG-expressing B cells from 4 individuals, we isolated 20 heterosubtypic mAbs that bound and neutralized viruses belonging to several HA subtypes (H1, H2, H5, H6, and H9), including the pandemic A/California/07/09 H1N1 isolate. The mAbs used different VH genes and carried a high frequency of somatic mutations. With the exception of a mAb that bound to the HA globular head, all heterosubtypic mAbs bound to acid-sensitive epitopes in the HA stem region. Four mAbs were evaluated in vivo and protected mice from challenge with influenza viruses representative of different subtypes. These findings reveal that seasonal influenza vaccination can induce polyclonal heterosubtypic neutralizing antibodies that cross-react with the swine-origin pandemic H1N1 influenza virus and with the highly pathogenic H5N1 virus.
Figures



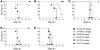

References
-
- Russell RJ, Burns WH, White DO, Anders EM, Ward CW, Jackson DC. Antigenic determinants of influenza virus hemagglutinin. III. Competitive binding of antibodies directed against “common” and “strain-specific” antigenic determinants of A/Memphis/72 hemagglutinin. J Immunol. 1979;123(2):825–832. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

