Ascorbic Acid inhibits development of tolerance and dependence to opiates in mice: possible glutamatergic or dopaminergic modulation
- PMID: 20390081
- PMCID: PMC2852062
- DOI: 10.4103/0250-474X.40332
Ascorbic Acid inhibits development of tolerance and dependence to opiates in mice: possible glutamatergic or dopaminergic modulation
Abstract
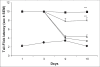
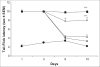
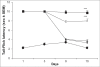
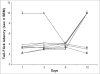
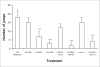
In a recent study, it has been demonstrated that ascorbic acid possessed antidopaminergic activity and modulate the glutamatergic neurotransmission in mice. With this background, the present study was undertaken to study the effect of ascorbic acid on the development of tolerance and dependence to opiate and its mechanism of action. Male Swiss mice weighing 20-25 g were used in the present study. Mice were made physically dependent on opioid by the chronic administration of morphine (10 mg/kg, twice a day, for 9 days) intraperitoneally. Ascorbic acid, haloperidol (dopamine antagonist) or MK 801 (NMDA receptor antagonist) was administered daily for 9 d before challenging the animals with morphine. The development of tolerance was assessed by noting the tail-flick latency on day 1, 3, 9 and 10. On the 10(th) day after the measurement of tail-flick latency, animals were challenged with naloxone (2 mg/kg., i.p.) and incidence of escape jumps were recorded by placing the animals in 45 cm high plexiglass container. Ascorbic acid (400-1600 mg/kg) dose dependently inhibited development of tolerance and dependence to morphine as noted from tail-flick latency. When given along with MK 801 (0.01 mg/kg., i.p) or haloperidol (0.1 mg/kg i.p.), ascorbic acid (800 mg/kg., i.p.) potentiated the response of MK 801 or haloperidol. In conclusion, it is hypothesized that inhibition of development of tolerance and dependence to morphine by ascorbic acid appears to have two components, namely dopaminergic and glutamatergic.
Keywords: Ascorbic acid; dependence; dopamine; glutamate; tolerance.
Figures
Similar articles
-
The importance of the number of NMDA receptors in the development of supersensitivity or tolerance to and dependence on morphine.Pharmacol Res. 1999 Apr;39(4):311-9. doi: 10.1006/phrs.1998.0443. Pharmacol Res. 1999. PMID: 10208762
-
Role of D1/D2 dopamine and N-methyl-D-aspartate (NMDA) receptors in morphine tolerance and dependence in mice.Eur Neuropsychopharmacol. 1995 Jun;5(2):81-7. doi: 10.1016/0924-977X(94)00140-7. Eur Neuropsychopharmacol. 1995. PMID: 7549459
-
Role of oxytocin in the neuroadaptation to drugs of abuse.Psychoneuroendocrinology. 1994;19(1):85-117. doi: 10.1016/0306-4530(94)90062-0. Psychoneuroendocrinology. 1994. PMID: 9210215 Review.
-
Attenuation of tolerance to opioid-induced antinociception and protection against morphine-induced decrease of neurofilament proteins by idazoxan and other I2-imidazoline ligands.Br J Pharmacol. 1998 Sep;125(1):175-85. doi: 10.1038/sj.bjp.0702031. Br J Pharmacol. 1998. PMID: 9776358 Free PMC article.
-
[Involvement of the amygdala on place aversion induced by naloxone in single-dose morphine-treated rats].Yakugaku Zasshi. 2008 Mar;128(3):395-403. doi: 10.1248/yakushi.128.395. Yakugaku Zasshi. 2008. PMID: 18311059 Review. Japanese.
Cited by
-
Ascorbic Acid interaction with analgesic effect of morphine and tramadol in mice.Anesth Pain Med. 2014 Jun 22;4(3):e19529. doi: 10.5812/aapm.19529. eCollection 2014 Aug. Anesth Pain Med. 2014. PMID: 25289375 Free PMC article.
-
Vitamin C, Pain and Opioid Use Disorder.Integr Med (Encinitas). 2020 Jun;19(3):18-29. Integr Med (Encinitas). 2020. PMID: 33132774 Free PMC article. Review.
References
-
- Bhargava HN. Diversity of agents that modify opioid tolerance, physical dependence, abstinence syndrome and self-administrative behavior. Pharmacol Rev. 1994;46:293–324. - PubMed
-
- Marek P, Ben-Eliyahu S, Gold M, Liebeskind JC. Excitatory amino acid antagonists (kynurenic acid and MK-801) attenuate the development of morphine tolerance in the rat. Brain Res. 1991;547:77–81. - PubMed
-
- Rasmussen K, Fuller RW, Stockton ME, Perry KW, Swinford RM, Ornstein PL. NMDA receptor antagonists suppress behaviors but not norepinephrine turnover or locus coeruleus unit activity induced by opiate withdrawal. Eur J Pharmacol. 1991;197:9–16. - PubMed
-
- Bhargava HN, Reddy PL, Gudehithlu KP. Down regulation of N-methyl-D-aspartate (NMDA) receptors of brain region and spinal cord of rats treated chronically with morphine. Gen Pharmacol. 1994;26:131–6. - PubMed
-
- Gudehithlu KP, Reddy PL, Bhargava HN. Effect of chronic morphine treatment on the binding of [3H] MK-801 to rat brain and spinal cord membranes. Pharmacologist. 1993;35:A190.
LinkOut - more resources
Full Text Sources
