Rett syndrome microglia damage dendrites and synapses by the elevated release of glutamate
- PMID: 20392956
- PMCID: PMC5533099
- DOI: 10.1523/JNEUROSCI.5966-09.2010
Rett syndrome microglia damage dendrites and synapses by the elevated release of glutamate
Abstract
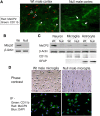
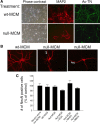
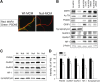
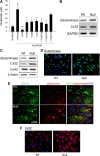
MECP2, an X-linked gene encoding the epigenetic factor methyl-CpG-binding protein-2, is mutated in Rett syndrome (RTT) and aberrantly expressed in autism. Most children affected by RTT are heterozygous Mecp2(-/+) females whose brain function is impaired postnatally due to MeCP2 deficiency. Recent studies suggest a role of glia in causing neuronal dysfunction via a non-cell-autonomous effect in RTT. Here we report a potent neurotoxic activity in the conditioned medium (CM) obtained from Mecp2-null microglia. Hippocampal neurons treated with CM from Mecp2-null microglia showed an abnormal stunted and beaded dendritic morphology, and signs of microtubule disruption and damage of postsynaptic glutamatergic components within 24 h. We identified that the toxic factor in the CM is glutamate, because (1) Mecp2-null microglia released a fivefold higher level of glutamate, (2) blockage of microglial glutamate synthesis by a glutaminase inhibitor abolished the neurotoxic activity, (3) blockage of microglial glutamate release by gap junction hemichannel blockers abolished the neurotoxic activity, and (4) glutamate receptor antagonists blocked the neurotoxicity of the Mecp2-null microglia CM. We further identified that increased levels of glutaminase and connexin 32 in Mecp2-null microglia are responsible for increased glutamate production and release, respectively. In contrast, the CM from highly pure Mecp2-null astrocyte cultures showed no toxic effect. Our results suggest that microglia may influence the onset and progression of RTT and that microglia glutamate synthesis or release could be a therapeutic target for RTT.
Figures
Similar articles
-
Rett syndrome astrocytes are abnormal and spread MeCP2 deficiency through gap junctions.J Neurosci. 2009 Apr 22;29(16):5051-61. doi: 10.1523/JNEUROSCI.0324-09.2009. J Neurosci. 2009. PMID: 19386901 Free PMC article.
-
Alterations of gene expression and glutamate clearance in astrocytes derived from an MeCP2-null mouse model of Rett syndrome.PLoS One. 2012;7(4):e35354. doi: 10.1371/journal.pone.0035354. Epub 2012 Apr 20. PLoS One. 2012. PMID: 22532851 Free PMC article.
-
Dendrimer-mediated delivery of N-acetyl cysteine to microglia in a mouse model of Rett syndrome.J Neuroinflammation. 2017 Dec 19;14(1):252. doi: 10.1186/s12974-017-1004-5. J Neuroinflammation. 2017. PMID: 29258545 Free PMC article.
-
Exploring the possible link between MeCP2 and oxidative stress in Rett syndrome.Free Radic Biol Med. 2015 Nov;88(Pt A):81-90. doi: 10.1016/j.freeradbiomed.2015.04.019. Epub 2015 May 8. Free Radic Biol Med. 2015. PMID: 25960047 Review.
-
Interpreting the rich dialogue between astrocytes and neurons: An overview in Rett syndrome.Brain Res Bull. 2025 Jul;227:111386. doi: 10.1016/j.brainresbull.2025.111386. Epub 2025 May 15. Brain Res Bull. 2025. PMID: 40378493 Review.
Cited by
-
X-chromosome inactivation in rett syndrome human induced pluripotent stem cells.Front Psychiatry. 2012 Mar 23;3:24. doi: 10.3389/fpsyt.2012.00024. eCollection 2012. Front Psychiatry. 2012. PMID: 22470355 Free PMC article.
-
IL-1β and TNF-α induce neurotoxicity through glutamate production: a potential role for neuronal glutaminase.J Neurochem. 2013 Jun;125(6):897-908. doi: 10.1111/jnc.12263. Epub 2013 May 3. J Neurochem. 2013. PMID: 23578284 Free PMC article.
-
Microglia and monocyte-derived macrophages: functionally distinct populations that act in concert in CNS plasticity and repair.Front Cell Neurosci. 2013 Apr 8;7:34. doi: 10.3389/fncel.2013.00034. eCollection 2013. Front Cell Neurosci. 2013. PMID: 23596391 Free PMC article.
-
How to reprogram microglia toward beneficial functions.Glia. 2018 Dec;66(12):2531-2549. doi: 10.1002/glia.23484. Epub 2018 Sep 8. Glia. 2018. PMID: 30195261 Free PMC article. Review.
-
A single-cell atlas reveals the heterogeneity of meningeal immunity in a mouse model of Methyl CpG binding protein 2 deficiency.Front Immunol. 2023 Jan 10;13:1056447. doi: 10.3389/fimmu.2022.1056447. eCollection 2022. Front Immunol. 2023. PMID: 36703978 Free PMC article.
References
-
- Anselmi F, Hernandez VH, Crispino G, Seydel A, Ortolano S, Roper SD, Kessaris N, Richardson W, Rickheit G, Filippov MA, Monyer H, Mammano F. ATP release through connexin hemichannels and gap junction transfer of second messengers propagate Ca2+ signals across the inner ear. Proc Natl Acad Sci U S A. 2008;105:18770–18775. - PMC - PubMed
-
- Armstrong DD. Neuropathology of Rett syndrome. J Child Neurol. 2005;20:747–753. - PubMed
-
- Asaka Y, Jugloff DG, Zhang L, Eubanks JH, Fitzsimonds RM. Hippocampal synaptic plasticity is impaired in the Mecp2-null mouse model of Rett syndrome. Neurobiol Dis. 2006;21:217–227. - PubMed
-
- Barger SW, Basile AS. Activation of microglia by secreted amyloid precursor protein evokes release of glutamate by cystine exchange and attenuates synaptic function. J Neurochem. 2001;76:846–854. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous
