Mitigation effect of an FGF-2 peptide on acute gastrointestinal syndrome after high-dose ionizing radiation
- PMID: 20394858
- PMCID: PMC2883168
- DOI: 10.1016/j.ijrobp.2009.11.026
Mitigation effect of an FGF-2 peptide on acute gastrointestinal syndrome after high-dose ionizing radiation
Abstract
Purpose: Acute gastrointestinal syndrome (AGS) resulting from ionizing radiation causes death within 7 days. Currently, no satisfactory agent exists for mitigation of AGS. A peptide derived from the receptor binding domain of fibroblast growth factor 2 (FGF-P) was synthesized and its mitigation effect on AGS was examined.
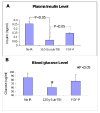
Methods and materials: A subtotal body irradiation (sub-TBI) model was created to induce gastrointestinal (GI) death while avoiding bone marrow death. After 10.5 to 16 Gy sub-TBI, mice received an intramuscular injection of FGF-P (10 mg/kg/day) or saline (0.2 ml/day) for 5 days; survival (frequency and duration) was measured. Crypt cells and their proliferation were assessed by hematoxylin, eosin, and BrdU staining. In addition, GI hemoccult score, stool formation, and plasma levels of endotoxin, insulin, amylase, interleukin (IL)-6, keratinocyte-derived chemokine (KC) monocyte chemoattractant protein 1 (MCP-1) and tumor necrosis factor (TNF)-alpha were evaluated.
Results: Treatment with FGF-P rescued a significant fraction of four strains of mice (33-50%) exposed to a lethal dose of sub-TBI. Use of FGF-P improved crypt survival and repopulation and partially preserved or restored GI function. Furthermore, whereas sub-TBI increased plasma endotoxin levels and several pro-inflammation cytokines (IL-6, KC, MCP-1, and TNF-alpha), FGF-P reduced these adverse responses.
Conclusions: The study data support pursuing FGF-P as a mitigator for AGS.
Conflict of interest statement
Figures





Similar articles
-
Post treatment with an FGF chimeric growth factor enhances epithelial cell proliferation to improve recovery from radiation-induced intestinal damage.Int J Radiat Oncol Biol Phys. 2010 Nov 1;78(3):860-7. doi: 10.1016/j.ijrobp.2010.04.045. Epub 2010 Aug 21. Int J Radiat Oncol Biol Phys. 2010. PMID: 20729008
-
Fibroblast growth factor-peptide improves barrier function and proliferation in human keratinocytes after radiation.Int J Radiat Oncol Biol Phys. 2011 Sep 1;81(1):248-54. doi: 10.1016/j.ijrobp.2011.02.004. Epub 2011 Apr 12. Int J Radiat Oncol Biol Phys. 2011. PMID: 21489707 Free PMC article.
-
Anti-ceramide Single-Chain Variable Fragment Mitigates Gastrointestinal-Acute Radiation Syndrome and Improves Marrow Reconstitution, Rendering Near-Normal 90-Day Autopsies.Int J Radiat Oncol Biol Phys. 2024 Oct 1;120(2):558-569. doi: 10.1016/j.ijrobp.2023.07.038. Epub 2023 Oct 9. Int J Radiat Oncol Biol Phys. 2024. PMID: 37815783 Free PMC article.
-
Modulation with cytokines of radiation injury: suggested mechanisms of action.Environ Health Perspect. 1997 Dec;105 Suppl 6(Suppl 6):1463-5. doi: 10.1289/ehp.97105s61463. Environ Health Perspect. 1997. PMID: 9467064 Free PMC article. Review.
-
Cyclophosphamide 24 hours before or after total body irradiation: effects on lung and bone marrow.Radiother Oncol. 1991 Jul;21(3):149-56. doi: 10.1016/0167-8140(91)90031-b. Radiother Oncol. 1991. PMID: 1924849 Review.
Cited by
-
Radiation damage and radioprotectants: new concepts in the era of molecular medicine.Br J Radiol. 2012 Apr;85(1012):313-30. doi: 10.1259/bjr/16386034. Epub 2012 Jan 31. Br J Radiol. 2012. PMID: 22294702 Free PMC article.
-
Medical Countermeasures for Radiation Exposure and Related Injuries: Characterization of Medicines, FDA-Approval Status and Inclusion into the Strategic National Stockpile.Health Phys. 2015 Jun;108(6):607-30. doi: 10.1097/HP.0000000000000279. Health Phys. 2015. PMID: 25905522 Free PMC article. Review.
-
Fibroblast Growth Factor Receptors as Targets for Radiosensitization in Head and Neck Squamous Cell Carcinomas.Int J Radiat Oncol Biol Phys. 2020 Jul 15;107(4):793-803. doi: 10.1016/j.ijrobp.2020.03.040. Epub 2020 Apr 13. Int J Radiat Oncol Biol Phys. 2020. PMID: 32298810 Free PMC article.
-
Therapeutic Potential of Chick Early Amniotic Fluid in Mitigating Ionizing-Radiation-Induced Damage.Biomedicines. 2025 May 21;13(5):1253. doi: 10.3390/biomedicines13051253. Biomedicines. 2025. PMID: 40427080 Free PMC article.
-
FGFR Inhibition Enhances Sensitivity to Radiation in Non-Small Cell Lung Cancer.Mol Cancer Ther. 2020 Jun;19(6):1255-1265. doi: 10.1158/1535-7163.MCT-19-0931. Epub 2020 May 5. Mol Cancer Ther. 2020. PMID: 32371583 Free PMC article.
References
-
- Freshwater DA. Effects of nuclear weapons on the gastrointestinal system. J R Army Med Corps. 2004;150(3 Suppl 1):17–21. - PubMed
-
- Ishii T, Futami S, Nishida M, et al. Brief note and evaluation of acute-radiation syndrome and treatment of a Tokai-mura criticality accident patient. J Radiat Res (Tokyo) 2001;42(Suppl):167–182. - PubMed
-
- Hensley ML, Hagerty KL, Kewalramani T, et al. American Society of Clinical Oncology 2008 clinical practice guideline update: use of chemotherapy and radiation therapy protectants. J Clin Oncol. 2009;27(1):127–145. - PubMed
-
- Cailliau K, Browaeys-Poly E, Vilain JP. Fibroblast growth factors 1 and 2 differently activate MAP kinase in Xenopus oocytes expressing fibroblast growth factor receptors 1 and 4. Biochem Biophys Acta. 2001;1538(2-3):228–233. - PubMed
-
- Diecke S, Quiroga-Negreira A, Redmer T, et al. FGF2 signaling in mouse embryonic fibroblasts is crucial for self-renewal of embryonic stem cells. Cells Tissues Organs. 2008;188(1-2):52–61. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

