Hyperglycaemia and diabetes impair gap junctional communication among astrocytes
- PMID: 20396375
- PMCID: PMC2839462
- DOI: 10.1042/AN20090048
Hyperglycaemia and diabetes impair gap junctional communication among astrocytes
Abstract
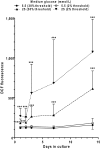
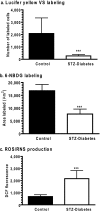
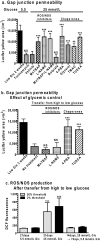
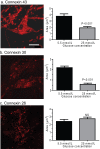
Sensory and cognitive impairments have been documented in diabetic humans and animals, but the pathophysiology of diabetes in the central nervous system is poorly understood. Because a high glucose level disrupts gap junctional communication in various cell types and astrocytes are extensively coupled by gap junctions to form large syncytia, the influence of experimental diabetes on gap junction channel-mediated dye transfer was assessed in astrocytes in tissue culture and in brain slices from diabetic rats. Astrocytes grown in 15-25 mmol/l glucose had a slow-onset, poorly reversible decrement in gap junctional communication compared with those grown in 5.5 mmol/l glucose. Astrocytes in brain slices from adult STZ (streptozotocin)-treated rats at 20-24 weeks after the onset of diabetes also exhibited reduced dye transfer. In cultured astrocytes grown in high glucose, increased oxidative stress preceded the decrement in dye transfer by several days, and gap junctional impairment was prevented, but not rescued, after its manifestation by compounds that can block or reduce oxidative stress. In sharp contrast with these findings, chaperone molecules known to facilitate protein folding could prevent and rescue gap junctional impairment, even in the presence of elevated glucose level and oxidative stress. Immunostaining of Cx (connexin) 43 and 30, but not Cx26, was altered by growth in high glucose. Disruption of astrocytic trafficking of metabolites and signalling molecules may alter interactions among astrocytes, neurons and endothelial cells and contribute to changes in brain function in diabetes. Involvement of the microvasculature may contribute to diabetic complications in the brain, the cardiovascular system and other organs.
Keywords: 4-PBA, 4-phenylbutyric acid; 6-NBDG, 6-[N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)amino]-2-deoxyglucose; Cx, connexin; DCF, 2′,7′-dichlorodihydrofluorescein; DIC, differential interference contrast; DMEM, Dulbecco's modified Eagle's medium; ER, endoplasmic reticulum; FBS, fetal bovine serum; LYCH, Lucifer Yellow CH; LYVS, Lucifer Yellow VS; MnTBAP, manganese(III) tetrakis (4-benzoic acid) porphyrin chloride; NA, numerical aperture; NOS, nitric oxide synthase; PKC, protein kinase C; RNS, reactive nitrogen species; ROS, reactive oxygen species; STZ, streptozotocin; TMAO, trimethylamine N-oxide dihydrate; TUDCA, tauroursodeoxycholic acid; aCSF, artificial cerebrospinal fluid; astrocyte; carboxy-DCF-DA, carboxy DCF diacetate; connexin (Cx); dBcAMP, dibutyryl cAMP; diabetes; gap junction; hyperglycaemia; l-NAME, l-Nω-nitro-l-arginine methyl ester; streptozotocin.
Figures




Similar articles
-
Astrocytic gap junctional communication is reduced in amyloid-β-treated cultured astrocytes, but not in Alzheimer's disease transgenic mice.ASN Neuro. 2010 Aug 17;2(4):e00041. doi: 10.1042/AN20100017. ASN Neuro. 2010. PMID: 20730033 Free PMC article.
-
Reduced gap junctional communication among astrocytes in experimental diabetes: contributions of altered connexin protein levels and oxidative-nitrosative modifications.J Neurosci Res. 2011 Dec;89(12):2052-67. doi: 10.1002/jnr.22663. Epub 2011 May 2. J Neurosci Res. 2011. PMID: 21567444 Free PMC article.
-
Involvement of gap junctions in astrocyte impairment induced by manganese exposure.Brain Res Bull. 2018 Jun;140:107-113. doi: 10.1016/j.brainresbull.2018.04.009. Epub 2018 Apr 17. Brain Res Bull. 2018. PMID: 29678775
-
Consequences of impaired gap junctional communication in glial cells.Adv Exp Med Biol. 1999;468:373-81. doi: 10.1007/978-1-4615-4685-6_29. Adv Exp Med Biol. 1999. PMID: 10635043 Review.
-
Connexin-based intercellular communication and astrocyte heterogeneity.Brain Res. 2012 Dec 3;1487:88-98. doi: 10.1016/j.brainres.2012.06.045. Epub 2012 Jul 10. Brain Res. 2012. PMID: 22789907 Review.
Cited by
-
Brain lactate metabolism: the discoveries and the controversies.J Cereb Blood Flow Metab. 2012 Jul;32(7):1107-38. doi: 10.1038/jcbfm.2011.175. Epub 2011 Dec 21. J Cereb Blood Flow Metab. 2012. PMID: 22186669 Free PMC article. Review.
-
Involvement of amylin B-H2S-connexin 43 signaling pathway in vascular dysfunction and enhanced ischemia-reperfusion-induced myocardial injury in diabetic rats.Biosci Rep. 2020 Jun 26;40(6):BSR20194154. doi: 10.1042/BSR20194154. Biosci Rep. 2020. PMID: 32436936 Free PMC article.
-
Effects of diabetes on brain metabolism--is brain glycogen a significant player?Metab Brain Dis. 2015 Feb;30(1):335-43. doi: 10.1007/s11011-014-9546-z. Epub 2014 Apr 29. Metab Brain Dis. 2015. PMID: 24771109 Review.
-
Connexin channel and its role in diabetic retinopathy.Prog Retin Eye Res. 2017 Nov;61:35-59. doi: 10.1016/j.preteyeres.2017.06.001. Epub 2017 Jun 8. Prog Retin Eye Res. 2017. PMID: 28602949 Free PMC article. Review.
-
Diabetes Mellitus and Blood-Brain Barrier Dysfunction: An Overview.J Pharmacovigil. 2014 Jun;2(2):125. doi: 10.4172/2329-6887.1000125. J Pharmacovigil. 2014. PMID: 25632404 Free PMC article.
References
-
- Abe T, Takahashi S, Suzuki N. Oxidative metabolism in cultured rat astroglia: effects of reducing the glucose concentration in the culture medium and of d-aspartate or potassium stimulation. J Cereb Blood Flow Metab. 2006;26:153–160. - PubMed
-
- Allen KV, Frier BM, Strachan MW. The relationship between type 2 diabetes and cognitive dysfunction: longitudinal studies and their methodological limitations. Eur J Pharmacol. 2004;490:169–175. - PubMed
-
- Beltramello M, Piazza V, Bukauskas FF, Pozzan T, Mammano F. Impaired permeability to Ins(1,4,5)P3 in a mutant connexin underlies recessive hereditary deafness. Nat Cell Biol. 2005;7:63–69. - PubMed
-
- Biessels GJ, Gispen WH. The impact of diabetes on cognition: what can be learned from rodent models? Neurobiol Aging. 2005;26 (Suppl. 1):36–41. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
