Spectroscopic and computational characterization of substrate-bound mouse cysteine dioxygenase: nature of the ferrous and ferric cysteine adducts and mechanistic implications
- PMID: 20397631
- PMCID: PMC2914100
- DOI: 10.1021/bi100189h
Spectroscopic and computational characterization of substrate-bound mouse cysteine dioxygenase: nature of the ferrous and ferric cysteine adducts and mechanistic implications
Abstract
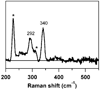
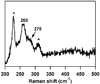
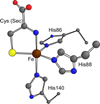
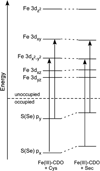
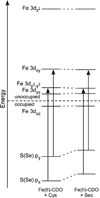
Cysteine dioxygenase (CDO) is a mononuclear non-heme Fe-dependent dioxygenase that catalyzes the initial step of oxidative cysteine catabolism. Its active site consists of an Fe(II) ion ligated by three histidine residues from the protein, an interesting variation on the more common 2-His-1-carboxylate motif found in many other non-heme Fe(II)-dependent enzymes. Multiple structural and kinetic studies of CDO have been carried out recently, resulting in a variety of proposed catalytic mechanisms; however, many open questions remain regarding the structure/function relationships of this vital enzyme. In this study, resting and substrate-bound forms of CDO in the Fe(II) and Fe(III) states, both of which are proposed to have important roles in this enzyme's catalytic mechanism, were characterized by utilizing various spectroscopic methods. The nature of the substrate/active site interactions was also explored using the cysteine analogue selenocysteine (Sec). Our electronic absorption, magnetic circular dichroism, and resonance Raman data exhibit features characteristic of direct S (or Se) ligation to both the high-spin Fe(II) and Fe(III) active site ions. The resulting Cys- (or Sec-) bound species were modeled and further characterized using density functional theory computations to generate experimentally validated geometric and electronic structure descriptions. Collectively, our results yield a more complete description of several catalytically relevant species and provide support for a reaction mechanism similar to that established for many structurally related 2-His-1-carboxylate Fe(II)-dependent dioxygenases.
Figures



References
-
- Yamaguchi K, Hosokawa Y. Cysteine dioxygenase. Methods Enzymol. 1987;143:395–403. - PubMed
-
- Lombardini JB, Singer TP, Boyer PD. Cysteine oxygenase. II. Studies on the mechanism of the reaction with 18oxygen. J. Biol. Chem. 1969;244:1172–1175. - PubMed
-
- Cooper AJL. Biochemistry of sulfur-containing amino acids. Annu. Rev. Biochem. 1983;52:187–222. - PubMed
-
- Stipanuk MH. Sulfur amino acid metabolism: pathways for production and removal of homocysteine and cysteine. Annu. Rev. Nutr. 2004;24:539–577. - PubMed
-
- Heafield MT, Fearn S, Steventon GB, Waring RH, Williams AC, Sturman SG. Plasma cysteine and sulphate levels in patients with motor neurone, Parkinson's and Alzheimer's disease. Neurosci. Lett. 1990;110:216–220. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

