Characterization of Fabry mice treated with recombinant adeno-associated virus 2/8-mediated gene transfer
- PMID: 20398385
- PMCID: PMC2861641
- DOI: 10.1186/1423-0127-17-26
Characterization of Fabry mice treated with recombinant adeno-associated virus 2/8-mediated gene transfer
Abstract
Background: Enzyme replacement therapy (ERT) with alpha-galactosidase A (alpha-Gal A) is currently the most effective therapeutic strategy for patients with Fabry disease, a lysosomal storage disease. However, ERT has limitations of a short half-life, requirement for frequent administration, and limited efficacy for patients with renal failure. Therefore, we investigated the efficacy of recombinant adeno-associated virus (rAAV) vector-mediated gene therapy for a Fabry disease mouse model and compared it with that of ERT.
Methods: A pseudotyped rAAV2/8 vector encoding alpha-Gal A cDNA (rAAV2/8-hAGA) was prepared and injected into 18-week-old male Fabry mice through the tail vein. The alpha-Gal A expression level and globotriaosylceramide (Gb3) levels in the Fabry mice were examined and compared with Fabry mice with ERT. Immunohistochemical and ultrastructural studies were conducted.
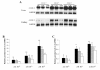
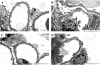
Results: Treatment of Fabry mice with rAAV2/8-hAGA resulted in the clearance of accumulated Gb3 in tissues such as liver, spleen, kidney, heart, and brain with concomitant elevation of alpha-Gal A enzyme activity. Enzyme activity was elevated for up to 60 weeks. In addition, expression of the alpha-Gal A protein was identified in the presence of rAAV2/8-hAGA at 6, 12, and 24 weeks after treatment. alpha-Gal A activity was significantly higher in the mice treated with rAAV2/8-hAGA than in Fabry mice that received ERT. Along with higher alpha-Gal A activity in the kidney of the Fabry mice treated with gene therapy, immunohistochemical studies showed more alpha-Gal A expression in the proximal tubules and glomerulus, and less Gb3 deposition in Fabry mice treated with this gene therapy than in mice given ERT. The alpha-gal A gene transfer significantly reduced the accumulation of Gb3 in the tubules and podocytes of the kidney. Electron microscopic analysis of the kidneys of Fabry mice also showed that gene therapy was more effective than ERT.
Conclusions: The rAAV2/8-hAGA mediated alpha-Gal A gene therapy provided improved efficiency over ERT in the Fabry disease mouse model. Furthermore, rAAV2/8-hAGA-mediated expression showed a greater effect in the kidney than ERT.
Figures
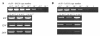



Similar articles
-
Long-term correction of globotriaosylceramide storage in Fabry mice by recombinant adeno-associated virus-mediated gene transfer.Proc Natl Acad Sci U S A. 2003 Mar 18;100(6):3450-4. doi: 10.1073/pnas.0537900100. Epub 2003 Mar 6. Proc Natl Acad Sci U S A. 2003. PMID: 12624185 Free PMC article.
-
Adeno-associated viral vector-mediated gene transfer results in long-term enzymatic and functional correction in multiple organs of Fabry mice.Proc Natl Acad Sci U S A. 2001 Feb 27;98(5):2676-81. doi: 10.1073/pnas.051634498. Proc Natl Acad Sci U S A. 2001. PMID: 11226298 Free PMC article.
-
Long-term inhibition of glycosphingolipid accumulation in Fabry model mice by a single systemic injection of AAV1 vector in the neonatal period.Mol Genet Metab. 2009 Mar;96(3):91-6. doi: 10.1016/j.ymgme.2008.10.017. Epub 2008 Dec 16. Mol Genet Metab. 2009. PMID: 19091614
-
Genetics and Gene Therapy of Anderson-Fabry Disease.Curr Gene Ther. 2018;18(2):96-106. doi: 10.2174/1566523218666180404161315. Curr Gene Ther. 2018. PMID: 29618309 Review.
-
Pathogenesis and Molecular Mechanisms of Anderson-Fabry Disease and Possible New Molecular Addressed Therapeutic Strategies.Int J Mol Sci. 2021 Sep 18;22(18):10088. doi: 10.3390/ijms221810088. Int J Mol Sci. 2021. PMID: 34576250 Free PMC article. Review.
Cited by
-
Fabry disease exacerbates renal interstitial fibrosis after unilateral ureteral obstruction via impaired autophagy and enhanced apoptosis.Kidney Res Clin Pract. 2021 Jun;40(2):208-219. doi: 10.23876/j.krcp.20.264. Epub 2021 May 21. Kidney Res Clin Pract. 2021. PMID: 34024086 Free PMC article.
-
Effects of enzyme replacement therapy in adult patients with Fabry disease on cardiac structure and function: a retrospective cohort study of the Fabry Munster Study (FaMuS) data.BMJ Open. 2012 Nov 21;2(6):bmjopen-2012-000879. doi: 10.1136/bmjopen-2012-000879. Print 2012. BMJ Open. 2012. PMID: 23175739 Free PMC article.
-
Effects of gene therapy on cardiovascular symptoms of lysosomal storage diseases.Genet Mol Biol. 2019;42(1 suppl 1):261-285. doi: 10.1590/1678-4685-GMB-2018-0100. Epub 2019 May 23. Genet Mol Biol. 2019. PMID: 31132295 Free PMC article.
-
Enhanced sialylation and in vivo efficacy of recombinant human α-galactosidase through in vitro glycosylation.BMB Rep. 2013 Mar;46(3):157-62. doi: 10.5483/bmbrep.2013.46.3.192. BMB Rep. 2013. PMID: 23527859 Free PMC article.
-
Sex differences of urinary and kidney globotriaosylceramide and lyso-globotriaosylceramide in Fabry mice.J Lipid Res. 2011 Sep;52(9):1742-6. doi: 10.1194/jlr.M017178. Epub 2011 Jul 11. J Lipid Res. 2011. PMID: 21747096 Free PMC article.
References
-
- Brady RO, Gal AE, Bradley RM, Martensson E, Warshaw AL, Laster L. Enzymatic defect in Fabry's disease: ceramidetrihexosidase deficiency. N Engl J Med. 1967;276:1163–1167. - PubMed
-
- Schiffmann R, Murray GJ, Treco D, Daniel P, Sellos-Moura M, Myers M, Quirk JM, Zirzow GC, Borowski M, Loveday K. Infusion of α-galactosidase A reduces tissue globotriaosylceramide storage in patients with Fabry disease. Proc Natl Acad Sci USA. 2000;97:365–370. doi: 10.1073/pnas.97.1.365. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

