Nucleoredoxin negatively regulates Toll-like receptor 4 signaling via recruitment of flightless-I to myeloid differentiation primary response gene (88)
- PMID: 20400501
- PMCID: PMC2881784
- DOI: 10.1074/jbc.M110.106468
Nucleoredoxin negatively regulates Toll-like receptor 4 signaling via recruitment of flightless-I to myeloid differentiation primary response gene (88)
Abstract
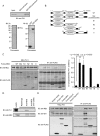
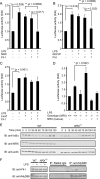
We previously characterized nucleoredoxin (NRX) as a negative regulator of the Wnt signaling pathway through Dishevelled (Dvl). We perform a comprehensive search for other NRX-interacting proteins and identify Flightless-I (Fli-I) as a novel NRX-binding partner. Fli-I binds to NRX and other related proteins, such as Rod-derived cone viability factor (RdCVF), whereas Dvl binds only to NRX. Endogenous NRX and Fli-I in vivo interactions are confirmed. Both NRX and RdCVF link Fli-I with myeloid differentiation primary response gene (88) (MyD88), an important adaptor protein for innate immune response. NRX and RdCVF also potentiate the negative effect of Fli-I upon lipopolysaccharide-induced activation of NF-kappaB through the Toll-like receptor 4/MyD88 pathway. Embryonic fibroblasts derived from NRX gene-targeted mice show aberrant NF-kappaB activation upon lipopolysaccharide stimulation. These results suggest that the NRX subfamily of proteins forms a link between MyD88 and Fli-I to mediate negative regulation of the Toll-like receptor 4/MyD88 pathway.
Figures



Similar articles
-
Nucleoredoxin sustains Wnt/β-catenin signaling by retaining a pool of inactive dishevelled protein.Curr Biol. 2010 Nov 9;20(21):1945-52. doi: 10.1016/j.cub.2010.09.065. Epub 2010 Oct 21. Curr Biol. 2010. PMID: 20970343
-
The thioredoxin-related redox-regulating protein nucleoredoxin inhibits Wnt-beta-catenin signalling through dishevelled.Nat Cell Biol. 2006 May;8(5):501-8. doi: 10.1038/ncb1405. Epub 2006 Apr 2. Nat Cell Biol. 2006. PMID: 16604061
-
Redox sensitivity of the MyD88 immune signaling adapter.Free Radic Biol Med. 2016 Dec;101:93-101. doi: 10.1016/j.freeradbiomed.2016.10.004. Epub 2016 Oct 5. Free Radic Biol Med. 2016. PMID: 27720842
-
Nucleoredoxin, a novel thioredoxin family member involved in cell growth and differentiation.Antioxid Redox Signal. 2007 Aug;9(8):1035-57. doi: 10.1089/ars.2007.1550. Antioxid Redox Signal. 2007. PMID: 17567240 Review.
-
Redox regulation of Wnt signalling via nucleoredoxin.Free Radic Res. 2010 Apr;44(4):379-88. doi: 10.3109/10715761003610745. Free Radic Res. 2010. PMID: 20187711 Review.
Cited by
-
Nucleoredoxin Knockdown in SH-SY5Y Cells Promotes Cell Renewal.Antioxidants (Basel). 2021 Mar 13;10(3):449. doi: 10.3390/antiox10030449. Antioxidants (Basel). 2021. PMID: 33805811 Free PMC article.
-
Is Nucleoredoxin a Master Regulator of Cellular Redox Homeostasis? Its Implication in Different Pathologies.Antioxidants (Basel). 2022 Mar 30;11(4):670. doi: 10.3390/antiox11040670. Antioxidants (Basel). 2022. PMID: 35453355 Free PMC article. Review.
-
Flightless-I is a potential biomarker for the early detection of alcoholic liver disease.Biochem Pharmacol. 2021 Jan;183:114323. doi: 10.1016/j.bcp.2020.114323. Epub 2020 Nov 7. Biochem Pharmacol. 2021. PMID: 33166508 Free PMC article.
-
Nucleoredoxin guards against oxidative stress by protecting antioxidant enzymes.Proc Natl Acad Sci U S A. 2017 Aug 1;114(31):8414-8419. doi: 10.1073/pnas.1703344114. Epub 2017 Jul 19. Proc Natl Acad Sci U S A. 2017. PMID: 28724723 Free PMC article.
-
Glutaredoxin 1 protects dopaminergic cells by increased protein glutathionylation in experimental Parkinson's disease.Antioxid Redox Signal. 2012 Dec 15;17(12):1676-93. doi: 10.1089/ars.2011.4474. Epub 2012 Sep 14. Antioxid Redox Signal. 2012. PMID: 22816731 Free PMC article.
References
-
- Lillig C. H., Holmgren A. (2007) Antioxid. Redox Signal. 9, 25–47 - PubMed
-
- Kurooka H., Kato K., Minoguchi S., Takahashi Y., Ikeda J., Habu S., Osawa N., Buchberg A. M., Moriwaki K., Shisa H., Honjo T. (1997) Genomics 39, 331–339 - PubMed
-
- Funato Y., Michiue T., Asashima M., Miki H. (2006) Nat. Cell Biol. 8, 501–508 - PubMed
-
- Clevers H. (2006) Cell 127, 469–480 - PubMed
-
- Funato Y., Michiue T., Terabayashi T., Yukita A., Danno H., Asashima M., Miki H. (2008) Genes Cells 13, 965–975 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

