CD4+ T cells elicit host immune responses to MHC class II-negative ovarian cancer through CCL5 secretion and CD40-mediated licensing of dendritic cells
- PMID: 20400704
- PMCID: PMC2874073
- DOI: 10.4049/jimmunol.0903247
CD4+ T cells elicit host immune responses to MHC class II-negative ovarian cancer through CCL5 secretion and CD40-mediated licensing of dendritic cells
Abstract
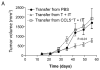
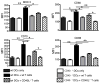
T cell adoptive transfer strategies that have produced clinical remissions against specific tumors have so far produced disappointing results against ovarian cancer. Recent evidence suggests that adoptively transferred CD4(+) T cells can trigger endogenous immune responses in particular patients with ovarian cancer through unknown mechanisms. However, conflicting reports suggest that ovarian cancer-infiltrating CD4(+) T cells are associated with negative outcomes. In this study, we elucidate the phenotypic attributes that enable polyclonal CD4(+) T cells briefly primed against tumor Ags to induce therapeutically relevant endogenous antitumor immune responses. Our results unveil a therapeutic mechanism whereby tumor-primed CD4(+) T cells transferred into ovarian cancer-bearing mice secrete high levels of CCL5, which recruits endogenous CCR5(+) dendritic cells to tumor locations and activate them through CD40-CD40L interactions. These newly matured dendritic cells are then able to prime tumor-specific endogenous CD8(+) T cells, which mediate long-term protection. Correspondingly, administration of tumor-primed CD4(+) T cells significantly delayed progression of MHC class II(-) ovarian cancers, similarly to CD8(+) T cells only, and directly activated wild-type but not CD40-deficient dendritic cells recruited to the tumor microenvironment. Our results unveil a CCL5- and CD40L-dependent mechanism of transferring immunity from exogenously activated CD4(+) T cells to tumor-exposed host cells, resulting in sustained antitumor effects. Our data provide a mechanistic rationale for incorporating tumor-reactive CD4(+) T cells in adoptive cell transfer immunotherapies against ovarian cancer and underscore the importance of optimizing immunotherapeutic strategies for the specific microenvironment of individual tumors.
Figures




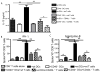
Similar articles
-
CCL5-mediated endogenous antitumor immunity elicited by adoptively transferred lymphocytes and dendritic cell depletion.Cancer Res. 2009 Aug 1;69(15):6331-8. doi: 10.1158/0008-5472.CAN-08-4329. Epub 2009 Jul 14. Cancer Res. 2009. PMID: 19602595 Free PMC article.
-
The duration of signaling through CD40 directs biological ability of dendritic cells to induce antitumor immunity.J Immunol. 2003 Dec 1;171(11):5828-36. doi: 10.4049/jimmunol.171.11.5828. J Immunol. 2003. PMID: 14634092
-
Role of CD4 T cell help and costimulation in CD8 T cell responses during Listeria monocytogenes infection.J Immunol. 2003 Feb 15;170(4):2053-63. doi: 10.4049/jimmunol.170.4.2053. J Immunol. 2003. PMID: 12574376
-
Central and effector memory CD4 and CD8 T-cell responses to tumor-associated antigens.Crit Rev Immunol. 2012;32(2):97-126. doi: 10.1615/critrevimmunol.v32.i2.10. Crit Rev Immunol. 2012. PMID: 23216610 Review.
-
Facts and Hopes of CD40 Agonists in Cancer Immunotherapy.Clin Cancer Res. 2025 Jun 3;31(11):2079-2087. doi: 10.1158/1078-0432.CCR-24-1660. Clin Cancer Res. 2025. PMID: 40117130 Review.
Cited by
-
Immunology and Immune Checkpoint Inhibition in Ovarian Cancer - Current Aspects.Geburtshilfe Frauenheilkd. 2021 Oct;81(10):1128-1144. doi: 10.1055/a-1475-4335. Epub 2021 Jul 6. Geburtshilfe Frauenheilkd. 2021. PMID: 34629492 Free PMC article.
-
Double Strike Approach for Tumor Attack: Engineering T Cells Using a CD40L:CD28 Chimeric Co-Stimulatory Switch Protein for Enhanced Tumor Targeting in Adoptive Cell Therapy.Front Immunol. 2021 Nov 29;12:750478. doi: 10.3389/fimmu.2021.750478. eCollection 2021. Front Immunol. 2021. PMID: 34912334 Free PMC article.
-
Upregulation of Apolipoprotein L6 Improves Tumor Immunotherapy by Inducing Immunogenic Cell Death.Biomolecules. 2023 Feb 22;13(3):415. doi: 10.3390/biom13030415. Biomolecules. 2023. PMID: 36979348 Free PMC article.
-
Anti-tumor immunity: myeloid leukocytes control the immune landscape.Cell Immunol. 2012 Jul-Aug;278(1-2):21-6. doi: 10.1016/j.cellimm.2012.06.014. Epub 2012 Jul 14. Cell Immunol. 2012. PMID: 23121972 Free PMC article. Review.
-
Therapeutic implications of the tumor microenvironment in ovarian cancer patients receiving PD-1/PD-L1 therapy.Front Immunol. 2022 Oct 20;13:1036298. doi: 10.3389/fimmu.2022.1036298. eCollection 2022. Front Immunol. 2022. PMID: 36341388 Free PMC article. Review.
References
-
- Zhang L, Conejo-Garcia JR, Katsaros D, Gimotty PA, Massobrio M, Regnani G, Makrigiannakis A, Gray H, Schlienger K, Liebman MN, Rubin SC, Coukos G. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N Engl J Med. 2003;348:203–213. - PubMed
-
- Sato E, Olson SH, Ahn J, Bundy B, Nishikawa H, Qian F, Jungbluth AA, Frosina D, Gnjatic S, Ambrosone C, Kepner J, Odunsi T, Ritter G, Lele S, Chen YT, Ohtani H, Old LJ, Odunsi K. Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proc Natl Acad Sci U S A. 2005;102:18538–18543. - PMC - PubMed
-
- Dudley ME, Wunderlich JR, Robbins PF, Yang JC, Hwu P, Schwartzentruber DJ, Topalian SL, Sherry R, Restifo NP, Hubicki AM, Robinson MR, Raffeld M, Duray P, Seipp CA, Rogers-Freezer L, Morton KE, Mavroukakis SA, White DE, Rosenberg SA. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science. 2002;298:850–854. - PMC - PubMed
-
- Dudley ME, Yang JC, Sherry R, Hughes MS, Royal R, Kammula U, Robbins PF, Huang J, Citrin DE, Leitman SF, Wunderlich J, Restifo NP, Thomasian A, Downey SG, Smith FO, Klapper J, Morton K, Laurencot C, White DE, Rosenberg SA. Adoptive cell therapy for patients with metastatic melanoma: evaluation of intensive myeloablative chemoradiation preparative regimens. J Clin Oncol. 2008;26:5233–5239. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

