Rapid and selective screening for sulfhydryl analytes in plasma and urine using surface-enhanced transmission mode desorption electrospray ionization mass spectrometry
- PMID: 20402469
- PMCID: PMC2872998
- DOI: 10.1021/ac100242b
Rapid and selective screening for sulfhydryl analytes in plasma and urine using surface-enhanced transmission mode desorption electrospray ionization mass spectrometry
Abstract
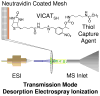
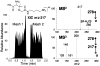
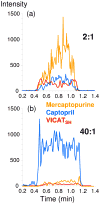
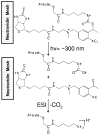
Nylon mesh substrates were derivatized to include VICAT(SH), a biotinylated reagent that contains both a photolabile linking group and a thiol specific capture agent. The enhanced mesh substrates were then used to capture sulfhydryl analytes directly from urine and plasma samples via covalent reaction between the reactive thiols of the analytes and the iodoacetaminyl unit of VICAT(SH). Photocleavage of the labile linker was followed by direct analysis of the mesh surface via transmission mode desorption electrospray ionization (TM-DESI). This chemoselective capture method promoted enrichment of sulfhydryl analytes and reduced matrix interferences, thereby resulting in increased analytical performance of surface enhanced TM-DESI-MS when compared to standard DESI-MS. The present work describes the manufacture of the derivatized mesh substrates and the quality control assessments made during the manufacturing process, the optimization of the chemoselective capture method, and results of experiments pertinent to biological applications. Integration of the chemoselective capture materials with ambient ionization and tandem mass spectrometry results in a powerful combination of speed and selectivity for targeted analyte screening.
Figures


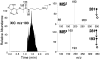


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

