Circulating monocytes from systemic sclerosis patients with interstitial lung disease show an enhanced profibrotic phenotype
- PMID: 20404807
- PMCID: PMC3682419
- DOI: 10.1038/labinvest.2010.73
Circulating monocytes from systemic sclerosis patients with interstitial lung disease show an enhanced profibrotic phenotype
Abstract
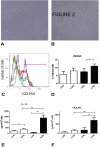
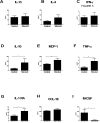
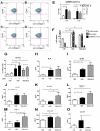
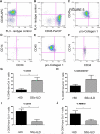
Profibrotic cells derived from circulating CD14+ monocytes include fibrocytes and alternatively activated macrophages. These cells are associated with interstitial lung disease (ILD) and are implicated in the pathogenesis of systemic sclerosis (SSc); however, the simultaneous presence of profibrotic cells and their associated mediators in the circulation of these patients has not been defined. We hypothesized that monocytes from patients with SSc-related ILD (SSc-ILD) would show profibrotic characteristics when compared with normal controls. We recruited patients with SSc-ILD (n=12) and normal controls (n=27) and quantified circulating collagen-producing cells by flow cytometry for CD45 and pro-collagen I. The in vitro activation potential of CD14+ monocytes in response to lipopolysaccharide was assessed using flow cytometry for CD163, and by ELISA for CCL18 and IL-10 secretion. Profibrotic mediators in plasma were quantified using Luminex-based assays. The concentration of circulating collagen-producing cells was increased in the SSc-ILD patients when compared with controls. These cells were composed of both CD34+ fibrocytes and a population of CD34+CD14+ cells. Cultured CD14+ monocytes from SSc-ILD patients revealed a profibrotic phenotype characterized by expression of CD163 and by enhanced secretion of CCL18 and IL-10 in response to proinflammatory activation. Plasma levels of IL-10, MCP-1, IL-1RA, and TNF levels were significantly elevated in the plasma of the SSc-ILD cohort. Subgroup analysis of the normal controls revealed that unlike the subjects < or =35 years, subjects > or =60 years old showed higher levels of circulating CD34+CD14+ cells, collagen-producing CD14+ monocytes, CD163+ monocytes, IL-4, IL-10, IL-13, MCP-1, and CCL18. These data indicate that the blood of patients with SSc-ILD and of healthy aged controls is enriched for fibrocytes, profibrotic monocytes, and fibrosis-associated mediators. Investigations defining the factors responsible for this peripheral blood profile may provide new insight into SSc-ILD as well as the pathophysiology of aging.
Figures

Similar articles
-
Prevalence of hybrid TLR4+M2 monocytes/macrophages in peripheral blood and lung of systemic sclerosis patients with interstitial lung disease.Front Immunol. 2024 Nov 20;15:1488867. doi: 10.3389/fimmu.2024.1488867. eCollection 2024. Front Immunol. 2024. PMID: 39635531 Free PMC article.
-
Netrin-1 Regulates Fibrocyte Accumulation in the Decellularized Fibrotic Sclerodermatous Lung Microenvironment and in Bleomycin-Induced Pulmonary Fibrosis.Arthritis Rheumatol. 2016 May;68(5):1251-61. doi: 10.1002/art.39575. Arthritis Rheumatol. 2016. PMID: 26749424 Free PMC article.
-
Elevated Fibronectin Levels in Profibrotic CD14+ Monocytes and CD14+ Macrophages in Systemic Sclerosis.Front Immunol. 2021 Aug 24;12:642891. doi: 10.3389/fimmu.2021.642891. eCollection 2021. Front Immunol. 2021. PMID: 34504485 Free PMC article.
-
Update on the pathogenesis of Scleroderma: focus on circulating progenitor cells.F1000Res. 2016 Apr 22;5:F1000 Faculty Rev-723. doi: 10.12688/f1000research.7986.1. eCollection 2016. F1000Res. 2016. PMID: 27158466 Free PMC article. Review.
-
Coagulation and autoimmunity in scleroderma interstitial lung disease.Semin Arthritis Rheum. 2011 Oct;41(2):212-22. doi: 10.1016/j.semarthrit.2010.10.002. Epub 2010 Dec 17. Semin Arthritis Rheum. 2011. PMID: 21168185 Free PMC article. Review.
Cited by
-
Migration of fibrocytes in fibrogenic liver injury.Am J Pathol. 2011 Jul;179(1):189-98. doi: 10.1016/j.ajpath.2011.03.049. Epub 2011 May 19. Am J Pathol. 2011. PMID: 21703401 Free PMC article.
-
Fibrocytes: emerging effector cells in chronic inflammation.Nat Rev Immunol. 2011 Jun;11(6):427-35. doi: 10.1038/nri2990. Epub 2011 May 20. Nat Rev Immunol. 2011. PMID: 21597472 Free PMC article.
-
Interleukin-1β and Tumor Necrosis Factor-α Gene Polymorphisms in Systemic Sclerosis.Balkan J Med Genet. 2025 Mar 6;27(2):59-68. doi: 10.2478/bjmg-2024-0017. eCollection 2024 Dec. Balkan J Med Genet. 2025. PMID: 40070856 Free PMC article.
-
Elevated Fcy receptor expression augments pro-inflammatory macrophage phagocytosis in systemic sclerosis and associated rheumatic diseases.Rheumatology (Oxford). 2025 Jun 1;64(6):3975-3988. doi: 10.1093/rheumatology/keae688. Rheumatology (Oxford). 2025. PMID: 39672802 Free PMC article.
-
Fibrocytes: emerging effector cells in chronic inflammation.Curr Opin Pharmacol. 2012 Aug;12(4):491-6. doi: 10.1016/j.coph.2012.03.002. Epub 2012 Mar 30. Curr Opin Pharmacol. 2012. PMID: 22465542 Free PMC article. Review.
References
-
- Tashkin DP, Elashoff R, Clements PJ, Goldin J, Roth MD, Furst DE, et al. Cyclophosphamide versus placebo in scleroderma lung disease. N Engl J Med. 2006;354(25):2655–66. - PubMed
-
- D'Ovidio F, Singer LG, Hadjiliadis D, Pierre A, Waddell TK, de Perrot M, et al. Prevalence of gastroesophageal reflux in end-stage lung disease candidates for lung transplant. Ann Thorac Surg. 2005;80(4):1254–60. - PubMed
-
- D'Ovidio F, Mura M, Tsang M, Waddell TK, Hutcheon MA, Singer LG, et al. Bile acid aspiration and the development of bronchiolitis obliterans after lung transplantation. J Thorac Cardiovasc Surg. 2005;129(5):1144–52. - PubMed
-
- Abe R, Donnelly SC, Peng T, Bucala R, Metz CN. Peripheral blood fibrocytes: differentiation pathway and migration to wound sites. J Immunol. 2001;166(12):7556–62. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

