Microglia-derived macrophage colony stimulating factor promotes generation of proinflammatory cytokines by astrocytes in the periventricular white matter in the hypoxic neonatal brain
- PMID: 20406232
- PMCID: PMC8094857
- DOI: 10.1111/j.1750-3639.2010.00387.x
Microglia-derived macrophage colony stimulating factor promotes generation of proinflammatory cytokines by astrocytes in the periventricular white matter in the hypoxic neonatal brain
Abstract
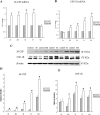
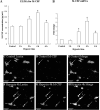
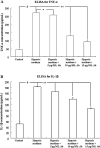
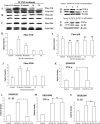
Inflammation in the periventricular white matter (PWM) of hypoxic neonatal brain causes myelination disturbances. In this connection, macrophage colony-stimulating factor (M-CSF) has been reported to regulate release of proinflammatory cytokines that may be linked to PWM damage. We sought to determine if M-CSF derived from amoeboid microglial cells (AMC) would promote proinflammatory cytokine production by astrocytes in the PWM following hypoxic exposure, and, if so, whether it is associated with axon degeneration and myelination disturbances. In 1-day hypoxic rats, expression of M-CSF was upregulated in AMC. This was coupled with increased expression of CSF-1 receptor, tumor necrosis factor-alpha (TNF-alpha) and interleukin-1beta (IL-1beta) in astrocytes, and TNF-receptor 1 and IL-receptor 1 on the axons. Neurofilament-200 immunopositive axons and myelin basic protein immunopositive processes appeared to undergo disruption in 14-days hypoxic rats. By electron microscopy, some axons showed degenerative changes affecting the microtubules and myelin sheath. Primary cultured microglial cells subjected to hypoxia showed enhanced release of M-CSF. Remarkably, primary cultured astrocytes treated with conditioned-medium derived from hypoxic microglia or M-CSF exhibited increased production of TNF-alpha and IL-1beta. Our results suggest that AMC-derived M-CSF promotes astrocytes to generate proinflammatory cytokines, which may be involved in axonal damage following a hypoxic insult.
Figures





References
-
- Barres BA, Raff MC (1993) Proliferation of oligodendrocyte precursor cells depends on electrical activity in axons. Nature 361:258–260. - PubMed
-
- Billiards SS, Haynes RL, Folkerth RD, Trachtenberg FL, Liu LG, Volpe JJ, Kinney HC (2006) Development of microglia in the cerebral white matter of the human fetus and infant. J Comp Neurol 497:199–208. - PubMed
-
- Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein‐dye binding. Anal Biochem 72:248–254. - PubMed
-
- Cecchini MG, Dominguez MG, Mocci S, Wetterwald A, Felix R, Fleisch H et al (1994) Role of colony stimulating factor‐1 in the establishment and regulation of tissue macrophages during postnatal development of the mouse. Development 120:1357–1372. - PubMed
-
- Chew LJ, Takanohashi A, Bell M (2006) Microglia and inflammation: impact on developmental brain injuries. Ment Retard Dev Disabil Res Rev 12:105–112. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

