Cetuximab, bevacizumab, and irinotecan for patients with primary glioblastoma and progression after radiation therapy and temozolomide: a phase II trial
- PMID: 20406901
- PMCID: PMC2940618
- DOI: 10.1093/neuonc/nop063
Cetuximab, bevacizumab, and irinotecan for patients with primary glioblastoma and progression after radiation therapy and temozolomide: a phase II trial
Abstract
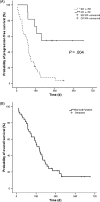
The aim of this clinical trial was to investigate safety and efficacy when combining cetuximab with bevacizumab and irinotecan in patients with recurrent primary glioblastoma multiforme (GBM). Patients were included with recurrent primary GBM and progression within 6 months of ending standard treatment (radiotherapy and temozolomide). Bevacizumab and irinotecan were administered IV every 2 weeks. The first 10 patients received bevacizumab 5 mg/kg, but this was increased to 10 mg/kg after interim safety analysis. Irinotecan dose was based on whether patients were taking enzyme-inducing antiepileptic drugs or not: 340 and 125 mg/m(2), respectively. Cetuximab 400 mg/m(2) as loading dose followed by 250 mg/m(2) weekly was administered IV. Forty-three patients were enrolled in the trial, of which 32 were available for response. Radiographic responses were noted in 34%, of which 2 patients had complete responses and 9 patients had partial responses. The 6-month progression-free survival probability was 30% and median overall survival was 29 weeks (95% CI: 23-37 weeks). One patient had lacunar infarction, 1 patient had multiple pulmonary embolisms, and 3 patients had grade 3 skin toxicity, for which 1 patient needed plastic surgery. One patient was excluded due to suspicion of interstitial lung disease. Three patients had deep-vein thrombosis; all continued on study after adequate treatment. Cetuximab in combination with bevacizumab and irinotecan in recurrent GBM is well tolerated except for skin toxicity, with an encouraging response rate. However, the efficacy data do not seem to be superior compared with results with bevacizumab and irinotecan alone.
Figures


Similar articles
-
Second-line chemotherapy with irinotecan plus carmustine in glioblastoma recurrent or progressive after first-line temozolomide chemotherapy: a phase II study of the Gruppo Italiano Cooperativo di Neuro-Oncologia (GICNO).J Clin Oncol. 2004 Dec 1;22(23):4779-86. doi: 10.1200/JCO.2004.06.181. J Clin Oncol. 2004. PMID: 15570079 Clinical Trial.
-
Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma.J Clin Oncol. 2009 Oct 1;27(28):4733-40. doi: 10.1200/JCO.2008.19.8721. Epub 2009 Aug 31. J Clin Oncol. 2009. PMID: 19720927 Clinical Trial.
-
Phase 2 study of carboplatin, irinotecan, and bevacizumab for recurrent glioblastoma after progression on bevacizumab therapy.Cancer. 2011 Dec 1;117(23):5351-8. doi: 10.1002/cncr.26188. Epub 2011 May 16. Cancer. 2011. PMID: 21590689 Free PMC article. Clinical Trial.
-
Bevacizumab: a treatment option for recurrent glioblastoma multiforme.Ann Pharmacother. 2008 Oct;42(10):1486-90. doi: 10.1345/aph.1L030. Epub 2008 Sep 2. Ann Pharmacother. 2008. PMID: 18765835 Review.
-
[Drug therapy of patients with recurrent glioblastoma: is there any evidence?].Wien Med Wochenschr. 2011 Jan;161(1-2):26-31. doi: 10.1007/s10354-010-0863-5. Epub 2010 Dec 27. Wien Med Wochenschr. 2011. PMID: 21181283 Review. German.
Cited by
-
Use of bevacizumab in recurrent glioblastoma.CNS Oncol. 2015;4(3):157-69. doi: 10.2217/cns.15.8. Epub 2015 Apr 23. CNS Oncol. 2015. PMID: 25906439 Free PMC article. Review.
-
Targeted Glioma Therapy-Clinical Trials and Future Directions.Pharmaceutics. 2024 Jan 11;16(1):100. doi: 10.3390/pharmaceutics16010100. Pharmaceutics. 2024. PMID: 38258110 Free PMC article. Review.
-
Chemotherapy for the treatment of intracranial glioma in dogs.Front Vet Sci. 2023 Oct 31;10:1273122. doi: 10.3389/fvets.2023.1273122. eCollection 2023. Front Vet Sci. 2023. PMID: 38026627 Free PMC article. Review.
-
Clinical benefit from resection of recurrent glioblastomas: results of a multicenter study including 503 patients with recurrent glioblastomas undergoing surgical resection.Neuro Oncol. 2016 Jan;18(1):96-104. doi: 10.1093/neuonc/nov145. Epub 2015 Aug 4. Neuro Oncol. 2016. PMID: 26243790 Free PMC article.
-
Angiotensinogen promoter methylation predicts bevacizumab treatment response of patients with recurrent glioblastoma.Mol Oncol. 2020 May;14(5):964-973. doi: 10.1002/1878-0261.12660. Epub 2020 Mar 18. Mol Oncol. 2020. PMID: 32133779 Free PMC article.
References
-
- Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352:987–996. - PubMed
-
- Wong ET, Hess KR, Gleason MJ, et al. Outcomes and prognostic factors in recurrent glioma patients enrolled onto phase II clinical trials. J Clin Oncol. 1999;17:2572–2578. - PubMed
-
- Vredenburgh JJ, Desjardins A, Herndon JE, 2nd, et al. Phase II trial of bevacizumab and irinotecan in recurrent malignant glioma. Clin Cancer Res. 2007;13:1253–1259. - PubMed
-
- Poulsen HS, Grunnet K, Sorensen M, et al. Bevacizumab plus irinotecan in the treatment patients with progressive recurrent malignant brain tumours. Acta Oncol. 2009;48:52–58. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

