Bestrophin-2 mediates bicarbonate transport by goblet cells in mouse colon
- PMID: 20407206
- PMCID: PMC2860923
- DOI: 10.1172/JCI41129
Bestrophin-2 mediates bicarbonate transport by goblet cells in mouse colon
Abstract
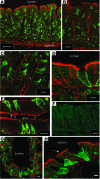
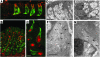
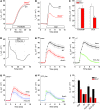
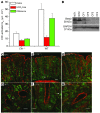
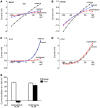
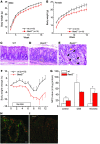
Anion transport by the colonic mucosa maintains the hydration and pH of the colonic lumen, and its disruption causes a variety of diarrheal diseases. Cholinergic agonists raise cytosolic Ca2+ levels and stimulate anion secretion, but the mechanisms underlying this effect remain unclear. Cholinergic stimulation of anion secretion may occur via activation of Ca2+-activated Cl- channels (CaCCs) or an increase in the Cl- driving force through CFTR after activation of Ca2+-dependent K+ channels. Here we investigated the role of a candidate CaCC protein, bestrophin-2 (Best2), using Best2-/- mice. Cholinergic stimulation of anion current was greatly reduced in Best2-/- mice, consistent with our proposed role for Best2 as a CaCC. However, immunostaining revealed Best2 localized to the basolateral membrane of mucin-secreting colonic goblet cells, not the apical membrane of Cl--secreting enterocytes. In addition, in the absence of HCO3-, cholinergic-activated current was identical in control and Best2-/- tissue preparations, which suggests that most of the Best2 current was carried by HCO3-. These data delineate an alternative model of cholinergic regulation of colonic anion secretion in which goblet cells play a critical role in HCO3- homeostasis. We therefore propose that Best2 is a HCO3- channel that works in concert with a Cl:HCO3- exchanger in the apical membrane to affect transcellular HCO3- transport. Furthermore, previous models implicating CFTR in cholinergic Cl- secretion may be explained by substantial downregulation of Best2 in Cftr-/- mice.
Figures




Similar articles
-
Lineage-specific expression of bestrophin-2 and bestrophin-4 in human intestinal epithelial cells.PLoS One. 2013 Nov 5;8(11):e79693. doi: 10.1371/journal.pone.0079693. eCollection 2013. PLoS One. 2013. PMID: 24223998 Free PMC article.
-
Bestrophin Cl- channels are highly permeable to HCO3-.Am J Physiol Cell Physiol. 2008 Jun;294(6):C1371-7. doi: 10.1152/ajpcell.00398.2007. Epub 2008 Apr 9. Am J Physiol Cell Physiol. 2008. PMID: 18400985 Free PMC article.
-
Bestrophin-2 is involved in the generation of intraocular pressure.Invest Ophthalmol Vis Sci. 2008 Apr;49(4):1563-70. doi: 10.1167/iovs.07-1338. Invest Ophthalmol Vis Sci. 2008. PMID: 18385076 Free PMC article.
-
Functional roles of bestrophins in ocular epithelia.Prog Retin Eye Res. 2009 May;28(3):206-26. doi: 10.1016/j.preteyeres.2009.04.004. Epub 2009 May 4. Prog Retin Eye Res. 2009. PMID: 19398034 Free PMC article. Review.
-
Mechanisms of cellular synchronization in the vascular wall. Mechanisms of vasomotion.Dan Med Bull. 2010 Oct;57(10):B4191. Dan Med Bull. 2010. PMID: 21040688 Review.
Cited by
-
Oncogenic potential of BEST4 in colorectal cancer via activation of PI3K/Akt signaling.Oncogene. 2022 Feb;41(8):1166-1177. doi: 10.1038/s41388-021-02160-2. Epub 2022 Jan 21. Oncogene. 2022. PMID: 35058597
-
Structural and functional characterization of the bestrophin-2 anion channel.Nat Struct Mol Biol. 2020 Apr;27(4):382-391. doi: 10.1038/s41594-020-0402-z. Epub 2020 Apr 6. Nat Struct Mol Biol. 2020. PMID: 32251414 Free PMC article.
-
Structure and Function of the Bestrophin family of calcium-activated chloride channels.Channels (Austin). 2021 Dec;15(1):604-623. doi: 10.1080/19336950.2021.1981625. Channels (Austin). 2021. PMID: 34612806 Free PMC article. Review.
-
Goblet cell LRRC26 regulates BK channel activation and protects against colitis in mice.Proc Natl Acad Sci U S A. 2021 Jan 19;118(3):e2019149118. doi: 10.1073/pnas.2019149118. Proc Natl Acad Sci U S A. 2021. PMID: 33431687 Free PMC article.
-
Eccrine sweat gland development and sweat secretion.Exp Dermatol. 2015 Sep;24(9):644-50. doi: 10.1111/exd.12773. Epub 2015 Jul 14. Exp Dermatol. 2015. PMID: 26014472 Free PMC article. Review.
References
-
- Kunzelmann K, Mall M. Electrolyte transport in the mammalian colon: mechanisms and implications for disease. Physiol Rev. 2002;82(1):245–289. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

