A cellulose synthase-like protein is required for osmotic stress tolerance in Arabidopsis
- PMID: 20409003
- PMCID: PMC3061338
- DOI: 10.1111/j.1365-313X.2010.04227.x
A cellulose synthase-like protein is required for osmotic stress tolerance in Arabidopsis
Abstract
Osmotic stress imposed by soil salinity and drought stress significantly affects plant growth and development, but osmotic stress sensing and tolerance mechanisms are not well understood. Forward genetic screens using a root-bending assay have previously identified salt overly sensitive (sos) mutants of Arabidopsis that fall into five loci, SOS1 to SOS5. These loci are required for the regulation of ion homeostasis or cell expansion under salt stress, but do not play a major role in plant tolerance to the osmotic stress component of soil salinity or drought. Here we report an additional sos mutant, sos6-1, which defines a locus essential for osmotic stress tolerance. sos6-1 plants are hypersensitive to salt stress and osmotic stress imposed by mannitol or polyethylene glycol in culture media or by water deficit in the soil. SOS6 encodes a cellulose synthase-like protein, AtCSLD5. Only modest differences in cell wall chemical composition could be detected, but we found that sos6-1 mutant plants accumulate high levels of reactive oxygen species (ROS) under osmotic stress and are hypersensitive to the oxidative stress reagent methyl viologen. The results suggest that SOS6/AtCSLD5 is not required for normal plant growth and development but has a critical role in osmotic stress tolerance and this function likely involves its regulation of ROS under stress.
Figures
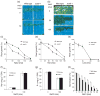
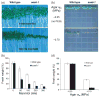
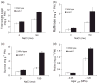


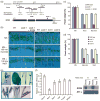
References
-
- Barkla BJ, Vera-Estrella R, Pantoja O. Towards the production of salt-tolerant crops. Adv Exp Med Biol. 1999;464:77–89. - PubMed
-
- Bernal AJ, Jensen JK, Harholt J, et al. Disruption of ATCSLD5 results in reduced growth, reduced xylan and homogalacturonan synthase activity and altered xylan occurrence in Arabidopsis. Plant J. 2007;52:791–802. - PubMed
-
- Blumenkrantz N, Asboe-Hansen G. New method for quantitative determination of uronic acids. Anal Biochem. 1973;54:484–489. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

