Mitochondrial oscillations and waves in cardiac myocytes: insights from computational models
- PMID: 20409461
- PMCID: PMC2856135
- DOI: 10.1016/j.bpj.2009.12.4300
Mitochondrial oscillations and waves in cardiac myocytes: insights from computational models
Abstract
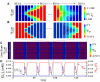
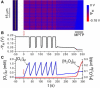
Periodic cellwide depolarizations of mitochondrial membrane potential (PsiM) which are triggered by reactive oxygen species (ROS) and propagated by ROS-induced ROS release (RIRR) have been postulated to contribute to cardiac arrhythmogenesis and injury during ischemia/reperfusion. Two different modes of RIRR have been described: PsiM oscillations involving ROS-sensitive mitochondrial inner membrane anion channels (IMAC), and slow depolarization waves related to mitochondrial permeability transition pore (MPTP) opening. In this study, we developed a computational model of mitochondria exhibiting both IMAC-mediated RIRR and MPTP-mediated RIRR, diffusively coupled in a spatially extended network, to study the spatiotemporal dynamics of RIRR on PsiM. Our major findings are: 1), as the rate of ROS production increases, mitochondria can exhibit either oscillatory dynamics facilitated by IMAC opening, or bistable dynamics facilitated by MPTP opening; 2), in a diffusively-coupled mitochondrial network, the oscillatory dynamics of IMAC-mediated RIRR results in rapidly propagating (approximately 25 microm/s) cellwide PsiM oscillations, whereas the bistable dynamics of MPTP-mediated RIRR results in slow (0.1-2 microm/s) PsiM depolarization waves; and 3), the slow velocity of the MPTP-mediated depolarization wave is related to competition between ROS scavenging systems and ROS diffusion. Our observations provide mechanistic insights into the spatiotemporal dynamics underlying RIRR-induced PsiM oscillations and waves observed experimentally in cardiac myocytes.
Copyright 2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures



References
-
- Stanley W.C., Recchia F.A., Lopaschuk G.D. Myocardial substrate metabolism in the normal and failing heart. Physiol. Rev. 2005;85:1093–1129. - PubMed
-
- Aon M.A., Cortassa S., O'Rourke B. Synchronized whole cell oscillations in mitochondrial metabolism triggered by a local release of reactive oxygen species in cardiac myocytes. J. Biol. Chem. 2003;278:44735–44744. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

