The measurement of lysosomal phospholipase A2 activity in plasma
- PMID: 20410020
- PMCID: PMC2903829
- DOI: 10.1194/jlr.D007146
The measurement of lysosomal phospholipase A2 activity in plasma
Abstract
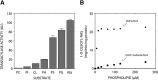
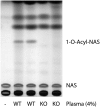
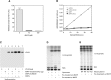
A deficiency of lysosomal phospholipase A2 (LPLA2) causes macrophage-associated phospholipidosis, suggesting that the enzyme is important in the lipid catabolism. Because LPLA2 is secreted by macrophages, extracellular LPLA2 activity may potentially reflect a change in macrophage activation. In this report, the detection of LPLA2 activity in plasma was established by the measurement of the transacylase activity of LPLA2 under acidic conditions. No transacylase activity of LPLA2 was detected in normal human plasma when the plasma was incubated with liposomes consisting of 1,2-dioleoylphosphatidylcholine/sulfatide/N-acetylsphingosine (NAS) at pH 4.5. However, the transacylase activity in the plasma was detected when liposomes consisting of 1,2-dioleoylphosphatidylglycerol/NAS were used as a substrate. To establish the specificity of the assay, ceramide transacylase activity was detected in the plasma of wild-type mice. By contrast, the plasma obtained from LPLA2-knockout mice had no measurable transacylase activity under the same conditions. The enzymatic activity of recombinant LPLA2 was inhibited by treatment with methylarachidonylfluorophosphonate. The inhibitor also suppressed the transacylase activity observed in both normal human and wild-type mouse plasma, establishing that the transacylase activity observed in plasma is due to LPLA2. Plasma LPLA2 activity may be a useful bioassay marker for the identification of LPLA2-related disorders.
Figures

Similar articles
-
Lysosomal phospholipase A2.Biochim Biophys Acta Mol Cell Biol Lipids. 2019 Jun;1864(6):932-940. doi: 10.1016/j.bbalip.2018.07.012. Epub 2018 Aug 2. Biochim Biophys Acta Mol Cell Biol Lipids. 2019. PMID: 30077006 Free PMC article. Review.
-
Preferential hydrolysis of truncated oxidized glycerophospholipids by lysosomal phospholipase A2.J Lipid Res. 2017 Feb;58(2):339-349. doi: 10.1194/jlr.M070730. Epub 2016 Dec 19. J Lipid Res. 2017. PMID: 27993948 Free PMC article.
-
A turn in the road: How studies on the pharmacology of glucosylceramide synthase inhibitors led to the identification of a lysosomal phospholipase A2 with ceramide transacylase activity.Glycoconj J. 2004;20(1):25-32. doi: 10.1023/B:GLYC.0000016739.32089.55. Glycoconj J. 2004. PMID: 14973367 Review.
-
Positional specificity of lysosomal phospholipase A2.J Lipid Res. 2006 Oct;47(10):2268-79. doi: 10.1194/jlr.M600183-JLR200. Epub 2006 Jul 12. J Lipid Res. 2006. PMID: 16837646
-
Esterification of side-chain oxysterols by lysosomal phospholipase A2.Biochim Biophys Acta Mol Cell Biol Lipids. 2020 Oct;1865(10):158787. doi: 10.1016/j.bbalip.2020.158787. Epub 2020 Aug 8. Biochim Biophys Acta Mol Cell Biol Lipids. 2020. PMID: 32777483
Cited by
-
NFATC3-PLA2G15 Fusion Transcript Identified by RNA Sequencing Promotes Tumor Invasion and Proliferation in Colorectal Cancer Cell Lines.Cancer Res Treat. 2019 Jan;51(1):391-401. doi: 10.4143/crt.2018.103. Epub 2018 Jun 14. Cancer Res Treat. 2019. PMID: 29909608 Free PMC article.
-
The phospholipase A2 activity of peroxiredoxin 6.J Lipid Res. 2018 Jul;59(7):1132-1147. doi: 10.1194/jlr.R082578. Epub 2018 May 1. J Lipid Res. 2018. PMID: 29716959 Free PMC article. Review.
-
Cellular and organismal function of choline metabolism.Nat Metab. 2025 Jan;7(1):35-52. doi: 10.1038/s42255-024-01203-8. Epub 2025 Jan 8. Nat Metab. 2025. PMID: 39779890 Review.
-
A fluorogenic phospholipid for the detection of lysosomal phospholipase A2 activity.Anal Biochem. 2013 Mar 1;434(1):78-83. doi: 10.1016/j.ab.2012.11.004. Epub 2012 Nov 9. Anal Biochem. 2013. PMID: 23146589 Free PMC article.
-
Lysosomal phospholipase A2.Biochim Biophys Acta Mol Cell Biol Lipids. 2019 Jun;1864(6):932-940. doi: 10.1016/j.bbalip.2018.07.012. Epub 2018 Aug 2. Biochim Biophys Acta Mol Cell Biol Lipids. 2019. PMID: 30077006 Free PMC article. Review.
References
-
- Abe A., Shayman J. A., Radin N. S. 1996. A novel enzyme that catalyzes the esterification of N-acetylsphingosine. Metabolism of C2-ceramides. J. Biol. Chem. 271: 14383–14389. - PubMed
-
- Abe A., Shayman J. A. 1998. Purification and characterization of 1-O-acylceramide synthase, a novel phospholipase A2 with transacylase activity. J. Biol. Chem. 273: 8467–8474. - PubMed
-
- Hiraoka M., Abe A., Shayman J. A. 2002. Cloning and characterization of a lysosomal phospholipase A2, 1-O-acylceramide synthase. J. Biol. Chem. 277: 10090–10099. - PubMed
-
- Schaloske R. H., Dennis E. A. 2006. The phospholipase A2 superfamily and its group numbering system. Biochim. Biophys. Acta. 1761: 1246–1259. - PubMed
-
- Abe A., Hiraoka M., Wild S., Wilcoxen S. E., Paine R., III, Shayman J. A. 2004. Lysosomal phospholipase A2 is selectively expressed in alveolar macrophages. J. Biol. Chem. 279: 42605–42611. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

