Metabolic vulnerability disposes retinal ganglion cell axons to dysfunction in a model of glaucomatous degeneration
- PMID: 20410117
- PMCID: PMC2884009
- DOI: 10.1523/JNEUROSCI.5956-09.2010
Metabolic vulnerability disposes retinal ganglion cell axons to dysfunction in a model of glaucomatous degeneration
Abstract
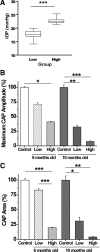
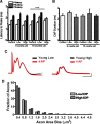
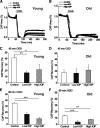
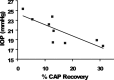
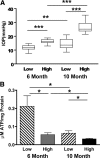
We tested the hypothesis that glaucoma disrupts electrophysiological conduction properties and axon function in optic nerve as a function of intraocular pressure (IOP) levels and age in the DBA/2J mouse model of glaucoma. The amplitude and the integral of electrical signals evoked along the axons decreased considerably by 6 months of age as a function of increasing IOP levels. At young ages, raised IOP was directly associated with increased vulnerability to metabolic challenge. Changes in the physiological function of the optic nerves were accentuated with aging, leading to loss of compound action potential in an entire population of fibers: small, slow conducting axons. This loss was accompanied with loss of small fiber axon counts and declining metabolic reserve by demonstrating IOP-dependent ATP decrease in mouse optic nerves. These data shed light on a novel potential mechanism of glaucoma pathology whereby increased IOP and declining metabolic capacity lead to axon liability and eventually dysfunction and loss.
Figures

References
-
- Anderson MG, Smith RS, Hawes NL, Zabaleta A, Chang B, Wiggs JL, John SW. Mutations in genes encoding melanosomal proteins cause pigmentary glaucoma in DBA/2J mice. Nat Genet. 2002;30:81–85. - PubMed
-
- Badea TC, Nathans J. Quantitative analysis of neuronal morphologies in the mouse retina visualized by using a genetically directed reporter. J Comp Neurol. 2004;480:331–351. - PubMed
-
- Baltan S, Inman DM, Danilov C, Morrison RS, Calkins DJ, Horner PJ. Reduced ATP levels dispose small size axons to dysfunction in DBA/2J glaucoma mice. Soc Neurosci Abstr. 2009;35:724–5.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
