Estradiol induction of spermatogenesis is mediated via an estrogen receptor-{alpha} mechanism involving neuroendocrine activation of follicle-stimulating hormone secretion
- PMID: 20410197
- PMCID: PMC2875821
- DOI: 10.1210/en.2009-1477
Estradiol induction of spermatogenesis is mediated via an estrogen receptor-{alpha} mechanism involving neuroendocrine activation of follicle-stimulating hormone secretion
Abstract
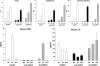
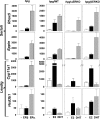
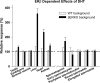
Both testosterone and its nonaromatizable metabolite dihydrotestosterone (DHT) induce spermatogenesis in gonadotropin-deficient hpg mice. Surprisingly, because aromatization is not required, estradiol (E2) also induces spermatogenesis and increases circulating FSH in hpg mice, but the mechanism remains unclear. We studied E2-induced spermatogenesis in hpg mice on an estrogen receptor (ER)-alpha (hpg/alphaERKO) or ERbeta (hpg/betaERKO) knockout or wild-type ER (hpg/WT) background treated with subdermal E2 or DHT implants for 6 wk. In hpg/WT and hpg/betaERKO, but not hpg/alphaERKO mice, E2 increased testis and epididymal weight, whereas DHT-induced increases were unaffected by ERalpha or ERbeta inactivation. E2 but not DHT treatment increased serum FSH (but not LH) in hpg/WT and hpg/betaERKO but not hpg/alphaERKO hpg mice. DHT or E2 alone increased (premeiotic) spermatogonia and (meiotic) spermatocytes without significant change in Sertoli cell numbers. DHT alone increased postmeiotic spermatids, regardless of ER presence, compared with variable ERalpha-dependent E2 postmeiotic responses. An ERalpha-mediated effect was confirmed by treating hpg mice for 6 wk by subdermal selective ER-alpha (16alpha-LE(2)) or ERbeta (8beta-VE(2)) agonist implants. ERalpha (but not ERbeta) agonist increased testis and epididymal weight, Sertoli cell, spermatogonia, meiotic, and postmeiotic germ cell numbers. Only ERalpha agonist markedly increased serum FSH, whereas either agonist induced small rises in serum LH. Administration of ERalpha agonist or E2 in the presence of functional ERalpha induced prominent gene expression of specific Sertoli (Eppin, Rhox5) and Leydig cell (Cyp11a1, Hsd3b1) markers. We conclude that E2-induced spermatogenesis in hpg mice involves an ERalpha-dependent neuroendocrine mechanism increasing blood FSH and Sertoli cell function.
Figures
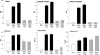


References
-
- O'Donnell L, Meachem SJ, Stanton PG, McLachlan RI 2006 Endocrine regulation of spermatogenesis. In: Neill JD, ed. Physiology of reproduction. 3rd ed. Amsterdam: Elsevier; 1017–1069
-
- Walker WH, Cheng J 2005 FSH and testosterone signaling in Sertoli cells. Reproduction 130:15–28 - PubMed
-
- Haywood M, Spaliviero J, Jimemez M, King NJ, Handelsman DJ, Allan CM 2003 Sertoli and germ cell development in hypogonadal (hpg) mice expressing transgenic follicle-stimulating hormone alone or in combination with testosterone. Endocrinology 144:509–517 - PubMed
-
- Orth JM, Gunsalus GL, Lamperti AA 1988 Evidence from Sertoli-cell depleted rats indicates that spermatid number depends on numbers of Sertoli cells produced during perinatal development. Endocrinology 122:787–794 - PubMed
-
- Russell LD, Ren HP, Sinha Hikim I, Schulze W, Sinha Hikim AP 1990 A comparative study in twelve mammalian species of volume densities, volumes, and numerical densities of selected testis components, emphasizing those related to the Sertoli cell. Am J Anat 188:21–30 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

