Xenin-25 potentiates glucose-dependent insulinotropic polypeptide action via a novel cholinergic relay mechanism
- PMID: 20421298
- PMCID: PMC2888395
- DOI: 10.1074/jbc.M110.129304
Xenin-25 potentiates glucose-dependent insulinotropic polypeptide action via a novel cholinergic relay mechanism
Abstract
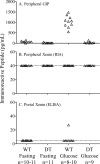
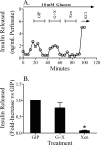
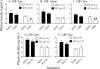
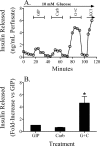
The intestinal peptides GLP-1 and GIP potentiate glucose-mediated insulin release. Agents that increase GLP-1 action are effective therapies in type 2 diabetes mellitus (T2DM). However, GIP action is blunted in T2DM, and GIP-based therapies have not been developed. Thus, it is important to increase our understanding of the mechanisms of GIP action. We developed mice lacking GIP-producing K cells. Like humans with T2DM, "GIP/DT" animals exhibited a normal insulin secretory response to exogenous GLP-1 but a blunted response to GIP. Pharmacologic doses of xenin-25, another peptide produced by K cells, restored the GIP-mediated insulin secretory response and reduced hyperglycemia in GIP/DT mice. Xenin-25 alone had no effect. Studies with islets, insulin-producing cell lines, and perfused pancreata indicated xenin-25 does not enhance GIP-mediated insulin release by acting directly on the beta-cell. The in vivo effects of xenin-25 to potentiate insulin release were inhibited by atropine sulfate and atropine methyl bromide but not by hexamethonium. Consistent with this, carbachol potentiated GIP-mediated insulin release from in situ perfused pancreata of GIP/DT mice. In vivo, xenin-25 did not activate c-fos expression in the hind brain or paraventricular nucleus of the hypothalamus indicating that central nervous system activation is not required. These data suggest that xenin-25 potentiates GIP-mediated insulin release by activating non-ganglionic cholinergic neurons that innervate the islets, presumably part of an enteric-neuronal-pancreatic pathway. Xenin-25, or molecules that increase acetylcholine receptor signaling in beta-cells, may represent a novel approach to overcome GIP resistance and therefore treat humans with T2DM.
Figures
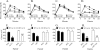

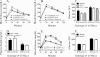
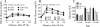
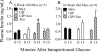




Similar articles
-
Xenin-25 amplifies GIP-mediated insulin secretion in humans with normal and impaired glucose tolerance but not type 2 diabetes.Diabetes. 2012 Jul;61(7):1793-800. doi: 10.2337/db11-1451. Epub 2012 Apr 20. Diabetes. 2012. PMID: 22522617 Free PMC article. Clinical Trial.
-
The combination of GIP plus xenin-25 indirectly increases pancreatic polypeptide release in humans with and without type 2 diabetes mellitus.Regul Pept. 2013 Nov 10;187:42-50. doi: 10.1016/j.regpep.2013.10.003. Epub 2013 Oct 29. Regul Pept. 2013. PMID: 24183983 Free PMC article.
-
A novel GLP-1/xenin hybrid peptide improves glucose homeostasis, circulating lipids and restores GIP sensitivity in high fat fed mice.Peptides. 2018 Feb;100:202-211. doi: 10.1016/j.peptides.2017.10.015. Peptides. 2018. PMID: 29412820
-
Glucose-dependent insulinotropic polypeptide: effects on insulin and glucagon secretion in humans.Dan Med J. 2016 Apr;63(4):B5230. Dan Med J. 2016. PMID: 27034187 Review.
-
Effects on pancreatic Beta and other Islet cells of the glucose-dependent insulinotropic polypeptide.Peptides. 2020 Mar;125:170201. doi: 10.1016/j.peptides.2019.170201. Epub 2019 Nov 18. Peptides. 2020. PMID: 31751656 Review.
Cited by
-
Inter-organ communication and regulation of beta cell function.Diabetologia. 2016 Apr;59(4):659-67. doi: 10.1007/s00125-015-3862-7. Epub 2016 Jan 20. Diabetologia. 2016. PMID: 26791990 Free PMC article. Review.
-
Glucose-dependent insulinotropic polypeptide (GIP).Mol Metab. 2025 May;95:102118. doi: 10.1016/j.molmet.2025.102118. Epub 2025 Feb 28. Mol Metab. 2025. PMID: 40024571 Free PMC article. Review.
-
Xenin-25 amplifies GIP-mediated insulin secretion in humans with normal and impaired glucose tolerance but not type 2 diabetes.Diabetes. 2012 Jul;61(7):1793-800. doi: 10.2337/db11-1451. Epub 2012 Apr 20. Diabetes. 2012. PMID: 22522617 Free PMC article. Clinical Trial.
-
Xenin and Related Peptides: Potential Therapeutic Role in Diabetes and Related Metabolic Disorders.Clin Med Insights Endocrinol Diabetes. 2021 Sep 22;14:11795514211043868. doi: 10.1177/11795514211043868. eCollection 2021. Clin Med Insights Endocrinol Diabetes. 2021. PMID: 34588834 Free PMC article. Review.
-
Neurotensin and its receptors in the control of glucose homeostasis.Front Endocrinol (Lausanne). 2012 Nov 26;3:143. doi: 10.3389/fendo.2012.00143. eCollection 2012. Front Endocrinol (Lausanne). 2012. PMID: 23230428 Free PMC article.
References
-
- Baggio L. L., Drucker D. J. (2007) Gastroenterology 132, 2131–2157 - PubMed
-
- Brand S. J., Schmidt W. E. (1995) in Textbook of Gastroenterology (Yamada T. ed) pp. 25–71, J. B. Lippincott Co., Philadelphia
-
- Fehmann H., Goke R., Goke B. (1995) Endocrine Rev. 16, 390–410 - PubMed
-
- Walsh J. H. (1994) in Physiology of the Gastrointestinal Tract (Johnson L. R. ed) pp. 1–128, Raven Press, New York
Publication types
MeSH terms
Substances
Grants and funding
- R37 DK031842/DK/NIDDK NIH HHS/United States
- P30 DK056341/DK/NIDDK NIH HHS/United States
- UL1 RR024992/RR/NCRR NIH HHS/United States
- RC1DK086163/DK/NIDDK NIH HHS/United States
- P60 DK020579/DK/NIDDK NIH HHS/United States
- P30 DK020579/DK/NIDDK NIH HHS/United States
- 5P30 DK052574/DK/NIDDK NIH HHS/United States
- DK31842/DK/NIDDK NIH HHS/United States
- UL1 TR000448/TR/NCATS NIH HHS/United States
- R01 DK088126/DK/NIDDK NIH HHS/United States
- R01 DK031842/DK/NIDDK NIH HHS/United States
- P30 DK052574/DK/NIDDK NIH HHS/United States
- RC1 DK086163/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

