Simulating microdosimetry in a virtual hepatic lobule
- PMID: 20421935
- PMCID: PMC2858695
- DOI: 10.1371/journal.pcbi.1000756
Simulating microdosimetry in a virtual hepatic lobule
Abstract
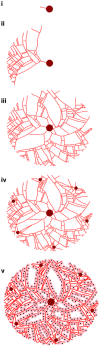
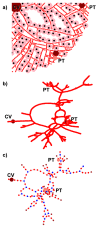
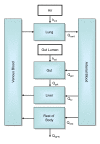
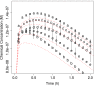
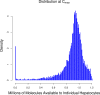
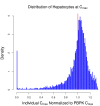
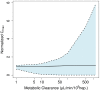
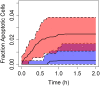
The liver plays a key role in removing harmful chemicals from the body and is therefore often the first tissue to suffer potentially adverse consequences. To protect public health it is necessary to quantitatively estimate the risk of long-term low dose exposure to environmental pollutants. Animal testing is the primary tool for extrapolating human risk but it is fraught with uncertainty, necessitating novel alternative approaches. Our goal is to integrate in vitro liver experiments with agent-based cellular models to simulate a spatially extended hepatic lobule. Here we describe a graphical model of the sinusoidal network that efficiently simulates portal to centrilobular mass transfer in the hepatic lobule. We analyzed the effects of vascular topology and metabolism on the cell-level distribution following oral exposure to chemicals. The spatial distribution of metabolically inactive chemicals was similar across different vascular networks and a baseline well-mixed compartment. When chemicals were rapidly metabolized, concentration heterogeneity of the parent compound increased across the vascular network. As a result, our spatially extended lobule generated greater variability in dose-dependent cellular responses, in this case apoptosis, than were observed in the classical well-mixed liver or in a parallel tubes model. The mass-balanced graphical approach to modeling the hepatic lobule is computationally efficient for simulating long-term exposure, modular for incorporating complex cellular interactions, and flexible for dealing with evolving tissues.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




Similar articles
-
Virtual tissues in toxicology.J Toxicol Environ Health B Crit Rev. 2010 Feb;13(2-4):314-28. doi: 10.1080/10937404.2010.483948. J Toxicol Environ Health B Crit Rev. 2010. PMID: 20574905
-
Modeling of xenobiotic transport and metabolism in virtual hepatic lobule models.PLoS One. 2018 Sep 13;13(9):e0198060. doi: 10.1371/journal.pone.0198060. eCollection 2018. PLoS One. 2018. PMID: 30212461 Free PMC article.
-
Computational systems biology and dose-response modeling in relation to new directions in toxicity testing.J Toxicol Environ Health B Crit Rev. 2010 Feb;13(2-4):253-76. doi: 10.1080/10937404.2010.483943. J Toxicol Environ Health B Crit Rev. 2010. PMID: 20574901
-
An overview of human exposure modeling activities at the USEPA's National Exposure Research Laboratory.Toxicol Ind Health. 2001 Jun;17(5-10):302-14. doi: 10.1191/0748233701th107oa. Toxicol Ind Health. 2001. PMID: 12539877 Review.
-
Molecular signaling network motifs provide a mechanistic basis for cellular threshold responses.Environ Health Perspect. 2014 Dec;122(12):1261-70. doi: 10.1289/ehp.1408244. Epub 2014 Aug 12. Environ Health Perspect. 2014. PMID: 25117432 Free PMC article. Review.
Cited by
-
Modeling drug- and chemical-induced hepatotoxicity with systems biology approaches.Front Physiol. 2012 Dec 14;3:462. doi: 10.3389/fphys.2012.00462. eCollection 2012. Front Physiol. 2012. PMID: 23248599 Free PMC article.
-
Hemodynamic flow improves rat hepatocyte morphology, function, and metabolic activity in vitro.Am J Physiol Cell Physiol. 2013 Jun 1;304(11):C1053-63. doi: 10.1152/ajpcell.00331.2012. Epub 2013 Mar 13. Am J Physiol Cell Physiol. 2013. PMID: 23485712 Free PMC article.
-
Mesoscale visualization of three-dimensional microvascular architecture and immunocyte distribution in intact mouse liver lobes.Theranostics. 2022 Jul 11;12(12):5418-5433. doi: 10.7150/thno.71718. eCollection 2022. Theranostics. 2022. PMID: 35910800 Free PMC article.
-
Liver Bioreactor Design Issues of Fluid Flow and Zonation, Fibrosis, and Mechanics: A Computational Perspective.J Funct Biomater. 2020 Feb 28;11(1):13. doi: 10.3390/jfb11010013. J Funct Biomater. 2020. PMID: 32121053 Free PMC article. Review.
-
Spatio-temporal simulation of first pass drug perfusion in the liver.PLoS Comput Biol. 2014 Mar 13;10(3):e1003499. doi: 10.1371/journal.pcbi.1003499. eCollection 2014 Mar. PLoS Comput Biol. 2014. PMID: 24625393 Free PMC article.
References
-
- Hartung T. Toxicology for the twenty-first century. Nature. 2009;460:208–212. - PubMed
-
- Krewski D, Andersen ME, Mantus E, Zeise L. Toxicity Testing in the 21st Century: Implications for Human Health Risk Assessment. Risk Analysis. 2007;29:474–479. - PubMed
-
- Dix DJ, Houck KA, Martin MT, Richard AM, Setzer RW, et al. The ToxCast program for prioritizing toxicity testing of environmental chemicals. Toxicological Sciences. 2007;95:5–12. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

