Comparison of the immune responses induced by chimeric alphavirus-vectored and formalin-inactivated alum-precipitated measles vaccines in mice
- PMID: 20421972
- PMCID: PMC2858653
- DOI: 10.1371/journal.pone.0010297
Comparison of the immune responses induced by chimeric alphavirus-vectored and formalin-inactivated alum-precipitated measles vaccines in mice
Abstract
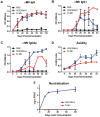
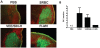
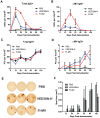
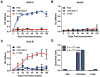
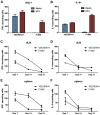
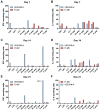
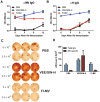
A variety of vaccine platforms are under study for development of new vaccines for measles. Problems with past measles vaccines are incompletely understood and underscore the need to understand the types of immune responses induced by different types of vaccines. Detailed immune response evaluation is most easily performed in mice. Although mice are not susceptible to infection with wild type or vaccine strains of measles virus, they can be used for comparative evaluation of the immune responses to measles vaccines of other types. In this study we compared the immune responses in mice to a new protective alphavirus replicon particle vaccine expressing the measles virus hemagglutinin (VEE/SIN-H) with a non-protective formalin-inactivated, alum-precipitated measles vaccine (FI-MV). MV-specific IgG levels were similar, but VEE/SIN-H antibody was high avidity IgG2a with neutralizing activity while FI-MV antibody was low-avidity IgG1 without neutralizing activity. FI-MV antibody was primarily against the nucleoprotein with no priming to H. Germinal centers appeared, peaked and resolved later for FI-MV. Lymph node MV antibody-secreting cells were more numerous after FI-MV than VEE/SIN-H, but were similar in the bone marrow. VEE/SIN-H-induced T cells produced IFN-gamma and IL-4 both spontaneously ex vivo and after stimulation, while FI-MV-induced T cells produced IL-4 only after stimulation. In summary, VEE/SIN-H induced a balanced T cell response and high avidity neutralizing IgG2a while FI-MV induced a type 2 T cell response, abundant plasmablasts, late germinal centers and low avidity non-neutralizing IgG1 against the nucleoprotein.
Conflict of interest statement
Figures

Similar articles
-
A chimeric alphavirus replicon particle vaccine expressing the hemagglutinin and fusion proteins protects juvenile and infant rhesus macaques from measles.J Virol. 2010 Apr;84(8):3798-807. doi: 10.1128/JVI.01566-09. Epub 2010 Feb 3. J Virol. 2010. PMID: 20130066 Free PMC article.
-
Modulation of disease, T cell responses, and measles virus clearance in monkeys vaccinated with H-encoding alphavirus replicon particles.Proc Natl Acad Sci U S A. 2005 Aug 16;102(33):11581-8. doi: 10.1073/pnas.0504592102. Epub 2005 Jul 21. Proc Natl Acad Sci U S A. 2005. PMID: 16037211 Free PMC article.
-
Neonatal immunization with a Sindbis virus-DNA measles vaccine induces adult-like neutralizing antibodies and cell-mediated immunity in the presence of maternal antibodies.J Immunol. 2006 May 1;176(9):5671-81. doi: 10.4049/jimmunol.176.9.5671. J Immunol. 2006. PMID: 16622037
-
Measles vaccines.Front Biosci. 2008 Jan 1;13:1352-70. doi: 10.2741/2767. Front Biosci. 2008. PMID: 17981635 Review.
-
Measles: old vaccines, new vaccines.Curr Top Microbiol Immunol. 2009;330:191-212. doi: 10.1007/978-3-540-70617-5_10. Curr Top Microbiol Immunol. 2009. PMID: 19203111 Review.
Cited by
-
Germinal Centre Shutdown.Front Immunol. 2021 Jul 7;12:705240. doi: 10.3389/fimmu.2021.705240. eCollection 2021. Front Immunol. 2021. PMID: 34305944 Free PMC article. Review.
-
Advanced vaccine candidates for Lassa fever.Viruses. 2012 Oct 29;4(11):2514-57. doi: 10.3390/v4112514. Viruses. 2012. PMID: 23202493 Free PMC article. Review.
-
Unusual patterns of IgG avidity in some young children following two doses of the adjuvanted pandemic H1N1 (2009) influenza virus vaccine.Clin Vaccine Immunol. 2013 Apr;20(4):459-67. doi: 10.1128/CVI.00619-12. Epub 2013 Jan 23. Clin Vaccine Immunol. 2013. PMID: 23345582 Free PMC article.
-
Lack of interference with immunogenicity of a chimeric alphavirus replicon particle-based influenza vaccine by preexisting antivector immunity.Clin Vaccine Immunol. 2012 Jul;19(7):991-8. doi: 10.1128/CVI.00031-12. Epub 2012 May 23. Clin Vaccine Immunol. 2012. PMID: 22623651 Free PMC article.
-
Generation of a More Immunogenic Measles Vaccine by Increasing Its Hemagglutinin Expression.J Virol. 2016 May 12;90(11):5270-5279. doi: 10.1128/JVI.00348-16. Print 2016 Jun 1. J Virol. 2016. PMID: 26984727 Free PMC article.
References
-
- Meissner HC, Strebel PM, Orenstein WA. Measles vaccines and the potential for worldwide eradication of measles. Pediatrics. 2004;114:1065–1069. - PubMed
-
- Albrecht P, Ennis FA, Saltzman EJ, Krugman S. Persistence of maternal antibody in infants beyond 12 months: Mechanism of measles vaccine failure. J Pediatr. 1977;91:715–178. - PubMed
-
- Gans HA, Arvin AM, Galinus J, Logan L, DeHovitz R, et al. Deficiency of the humoral immune response to measles vaccine in infants immunized at age 6 months. JAMA. 1998;280:527–532. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

