A role for the Kolliker-Fuse nucleus in cholinergic modulation of breathing at night during wakefulness and NREM sleep
- PMID: 20431024
- PMCID: PMC2904195
- DOI: 10.1152/japplphysiol.00933.2009
A role for the Kolliker-Fuse nucleus in cholinergic modulation of breathing at night during wakefulness and NREM sleep
Abstract
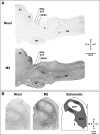
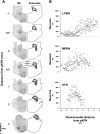
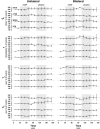
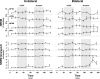
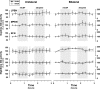
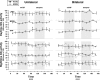
For many years, acetylcholine has been known to contribute to the control of breathing and sleep. To probe further the contributions of cholinergic rostral pontine systems in control of breathing, we designed this study to test the hypothesis that microdialysis (MD) of the muscarinic receptor antagonist atropine into the pontine respiratory group (PRG) would decrease breathing more in animals while awake than while in NREM sleep. In 16 goats, cannulas were bilaterally implanted into rostral pontine tegmental nuclei (n = 3), the lateral (n = 3) or medial (n = 4) parabrachial nuclei, or the Kölliker-Fuse nucleus (KFN; n = 6). After >2 wk of recovery from surgery, the goats were studied during a 45-min period of MD with mock cerebrospinal fluid (mCSF), followed by at least 30 min of recovery and a second 45-min period of MD with atropine. Unilateral and bilateral MD studies were completed during the day and at night. MD of atropine into the KFN at night decreased pulmonary ventilation and breathing frequency and increased inspiratory and expiratory time by 12-14% during both wakefulness and NREM sleep. However, during daytime studies, MD of atropine into the KFN had no effect on these variables. Unilateral and bilateral nighttime MD of atropine into the KFN increased levels of NREM sleep by 63 and 365%, respectively. MD during the day or at night into the other three pontine sites had minimal effects on any variable studied. Finally, compared with MD of mCSF, bilateral MD of atropine decreased levels of acetylcholine and choline in the effluent dialysis fluid. Our data support the concept that the KFN is a significant contributor to cholinergically modulated control of breathing and sleep.
Figures


References
-
- Baghdoyan H, Lydic R, Callaway C, Hobson J. The carbachol-induced enhancement of desynchronized sleep signs is dose dependent and antagonized by centrally administered atropine. Neuropsychopharmacology 2: 67– 79, 1989 - PubMed
-
- Baghdoyan H, Monaco A, Rodrigo-Angulo M, Assens F, McCarley R, Hobson J. Microinjection of neostigmine into the pontine reticular formation of cats enhances desynchronized sleep signs. J Pharmacol Exp Ther 231: 173– 180, 1984 - PubMed
-
- Bagydoyan H, Rodrigo-Angulo M, McCarley R, Hobson J. Site-specific enhancement and suppression of desynchronized sleep signs following cholinergic stimulation of three brainstem regions. Brain Res 306: 39– 52, 1984 - PubMed
-
- Barassin S, Raison S, Saboureau M, Bienvenu C, Maitre M, Malan A, Pevet P. Circadian tryptophan hydroxylase levels and serotonin release in the suprachiasmatic nucleus of the rat. Eur J Neurosci 15: 833– 840, 2002 - PubMed
-
- Bellingham MC, Berger AJ. Presynaptic depression of excitatory synaptic inputs to rat hypoglossal motoneurons by muscarinic M2 receptors. J Neurophysiol 76: 3758– 3770, 1996 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

