Generation of endoderm-derived human induced pluripotent stem cells from primary hepatocytes
- PMID: 20432258
- PMCID: PMC2925460
- DOI: 10.1002/hep.23626
Generation of endoderm-derived human induced pluripotent stem cells from primary hepatocytes
Abstract
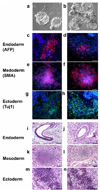
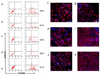
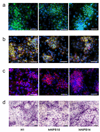
Recent advances in induced pluripotent stem (iPS) cell research have significantly changed our perspective on regenerative medicine. Patient-specific iPS cells have been derived not only for disease modeling but also as sources for cell replacement therapy. However, there have been insufficient data to prove that iPS cells are functionally equivalent to human embryonic stem (hES) cells or are safer than hES cells. There are several important issues that need to be addressed, and foremost are the safety and efficacy of human iPS cells of different origins. Human iPS cells have been derived mostly from cells originating from mesoderm and in a few cases from ectoderm. So far, there has been no report of endoderm-derived human iPS cells, and this has prevented comprehensive comparative investigations of the quality of human iPS cells of different origins. Here we show for the first time reprogramming of human endoderm-derived cells (i.e., primary hepatocytes) to pluripotency. Hepatocyte-derived iPS cells appear indistinguishable from hES cells with respect to colony morphology, growth properties, expression of pluripotency-associated transcription factors and surface markers, and differentiation potential in embryoid body formation and teratoma assays. In addition, these cells are able to directly differentiate into definitive endoderm, hepatic progenitors, and mature hepatocytes.
Conclusion: The technology to develop endoderm-derived human iPS cell lines, together with other established cell lines, will provide a foundation for elucidating the mechanisms of cellular reprogramming and for studying the safety and efficacy of differentially originated human iPS cells for cell therapy. For the study of liver disease pathogenesis, this technology also provides a potentially more amenable system for generating liver disease-specific iPS cells.
Figures


Comment in
-
Generation of human induced pluripotent stem cells from liver progenitor cells by only small molecules.Hepatology. 2010 Sep;52(3):1169; author reply 1169-70. doi: 10.1002/hep.23851. Hepatology. 2010. PMID: 20812362 No abstract available.
References
-
- Miura K, Okada Y, Aoi T, Okada A, Takahashi K, Okita K, et al. Variation in the safety of induced pluripotent stem cell lines. Nat Biotechnol. 2009;27:743–745. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
