Mechanistic and signaling analysis of Muc4-ErbB2 signaling module: new insights into the mechanism of ligand-independent ErbB2 activity
- PMID: 20432461
- PMCID: PMC2897936
- DOI: 10.1002/jcp.22163
Mechanistic and signaling analysis of Muc4-ErbB2 signaling module: new insights into the mechanism of ligand-independent ErbB2 activity
Abstract
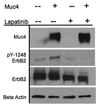
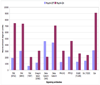
The membrane mucin Muc4 is aberrantly expressed in numerous epithelial carcinomas and is currently used as a cancer diagnostic and prognostic tool. Muc4 can also potentiate signal transduction by modulating differential ErbB2 phosphorylation in the absence and in the presence of the ErbB3 soluble ligand heregulin (HRG-beta1). These features of Muc4 suggest that Muc4 is not merely a cancer marker, but an oncogenic factor with a unique-binding/activation relationship with the receptor ErbB2. In the present study, we examined the signaling mechanisms that are associated with the Muc4-ErbB2 module by analyzing ErbB2 differential signaling in response to Muc4 expression. Our study was carried out in the A375 human melanoma and BT-474 breast cancer cell lines as our model systems. Quantitative and comparative signaling modulations were evaluated by immunoblot using phospho-specific antibodies, and densitometry analysis. Signaling complex components were identified by chemical cross-linking, fractionation by gel filtration, immunoprecipitation, and immunoblotting. Activated downstream signaling pathways were analyzed by an antibody microarray screen and immunoblot analyses. Our results indicate that Muc4 modulates ErbB2 signaling potential significantly by stabilizing and directly interacting with the ErbB2-ErbB3 heterodimer. Further analyses indicate that Muc4 promotes ErbB2 autocatalysis, but it has no effect on ErbB3 phosphorylation, although the chemical cross-linking data indicated that the signaling module is composed of Muc4, ErbB2, and ErbB3. Our microarray analysis indicates that Muc4 expression promotes cell migration by increasing the phosphorylation of the focal adhesion kinase and also through an increase in the levels of beta-catenin.
(c) 2010 Wiley-Liss, Inc.
Figures







Similar articles
-
MUC4 involvement in ErbB2/ErbB3 phosphorylation and signaling in response to airway cell mechanical injury.J Cell Biochem. 2009 May 1;107(1):112-22. doi: 10.1002/jcb.22106. J Cell Biochem. 2009. PMID: 19288496
-
The membrane mucin Muc4 inhibits apoptosis induced by multiple insults via ErbB2-dependent and ErbB2-independent mechanisms.Cancer Res. 2009 Apr 1;69(7):2845-52. doi: 10.1158/0008-5472.CAN-08-2089. Epub 2009 Mar 17. Cancer Res. 2009. PMID: 19293191 Free PMC article.
-
Muc4-ErbB2 complex formation and signaling in polarized CACO-2 epithelial cells indicate that Muc4 acts as an unorthodox ligand for ErbB2.Mol Biol Cell. 2006 Jul;17(7):2931-41. doi: 10.1091/mbc.e05-09-0895. Epub 2006 Apr 19. Mol Biol Cell. 2006. PMID: 16624867 Free PMC article.
-
Muc4/MUC4 functions and regulation in cancer.Future Oncol. 2009 Dec;5(10):1631-40. doi: 10.2217/fon.09.125. Future Oncol. 2009. PMID: 20001800 Free PMC article. Review.
-
Cell signaling through membrane mucins.Bioessays. 2003 Jan;25(1):66-71. doi: 10.1002/bies.10201. Bioessays. 2003. PMID: 12508284 Review.
Cited by
-
Cell membrane-anchored MUC4 promotes tumorigenicity in epithelial carcinomas.Oncotarget. 2017 Feb 21;8(8):14147-14157. doi: 10.18632/oncotarget.13122. Oncotarget. 2017. PMID: 27829225 Free PMC article. Review.
-
Palmitoylation: a protein S-acylation with implications for breast cancer.NPJ Breast Cancer. 2016 Oct 19;2:16028. doi: 10.1038/npjbcancer.2016.28. eCollection 2016. NPJ Breast Cancer. 2016. PMID: 28721385 Free PMC article. Review.
-
The multifaceted roles of mucins family in lung cancer: from prognostic biomarkers to promising targets.Front Immunol. 2025 Jun 27;16:1608140. doi: 10.3389/fimmu.2025.1608140. eCollection 2025. Front Immunol. 2025. PMID: 40655139 Free PMC article. Review.
-
Epigenetic control of HNF-4α in colon carcinoma cells affects MUC4 expression and malignancy.Cell Oncol (Dordr). 2013 Apr;36(2):155-67. doi: 10.1007/s13402-012-0123-3. Epub 2013 Jan 11. Cell Oncol (Dordr). 2013. PMID: 23307400
-
Assessing the prognostic significance of MUC4β in mucoepidermoid carcinoma of the salivary glands: An immunohistochemical study.Heliyon. 2019 Nov 14;5(11):e02753. doi: 10.1016/j.heliyon.2019.e02753. eCollection 2019 Nov. Heliyon. 2019. PMID: 31844699 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

