Management and monitoring of anticoagulation for children undergoing cardiopulmonary bypass in cardiac surgery
- PMID: 20437787
- PMCID: PMC4680072
Management and monitoring of anticoagulation for children undergoing cardiopulmonary bypass in cardiac surgery
Abstract
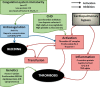
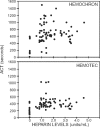
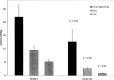
Cardiopulmonary bypass (CPB) creates a pro-coagulant state by causing platelet activation and inflammation leading to thrombin generation and platelet dysfunction. It is associated with severe derangements in normal homeostasis resulting in both thrombotic and hemorrhagic complications. This derangement is greater in children with congenital heart disease than in adults because of the immaturity of the coagulation system, hemodilution of coagulation factors, hyperreactive platelets, and in some patients, physiologic changes associated with cyanosis. During CPB, an appropriate amount of heparin is given with the goal of minimizing the risk of thrombosis and platelet activation and at the same time reducing the risk of bleeding from over anticoagulation. In young children, this balance is more difficult to achieve because of inherent characteristics of the hemostatic system in these patients. Historically, protocols for heparin dosing and monitoring in children have been adapted from adult protocols without re-validation for children. Extreme hemodilution of coagulation factors and platelets in young children affects the accuracy of anticoagulation monitoring in children. The activated clotting time does not correlate with plasma levels of heparin. In addition, recent studies suggest that children need larger doses of heparin than adults, because they have lower antithrombin levels, and they metabolize heparin more rapidly. Preliminary studies demonstrated that the use of individualized heparin and protamine monitoring and management in children is associated with reduced platelet activation and dysfunction and improved clinical outcomes. However, this review article clearly establishes that further studies are necessary to obtain evidence-based protocols for the proper management of anticoagulation of children undergoing cardiopulmonary bypass.
Conflict of interest statement
The senior author has stated that authors have reported no material, financial, or other relationship with any healthcare-related business or other entity whose products or services are discussed in this paper.
Figures
References
-
- Monagle P.. Thrombosis in children with BT shunts, Glenns and Fontans. Prog Pediatr Cardiol. 2005;21:17–21.
-
- Cholette JM, Rubenstein JS, Alfieris GM, et al. . Elevated risk of thrombosis in neonates undergoing initial palliative cardiac surgery. Ann Thorac Surg. 2007;84:1320–5. - PubMed
-
- Monagle P, Barnes C, Ignjatovic V, et al. . Developmental haemostasis. Impact for clinical haemostasis laboratories. Thromb Haemost. 2006;95:362–72. - PubMed
-
- Andrew M, Vegh P, Johnston M, Bowker J, Ofosu F, Mitchell L.. Maturation of the hemostatic system during childhood. Blood. 1992;80:1998–2005. - PubMed
-
- Svenmarker S, Appelblad M, Jansson E, Haggmark S.. Measurement of the activated clotting time during cardiopulmonary bypass: Differences between HemoTec ACT and Hemochron Jr apparatus. Perfusion. 2004;19:289–94. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
