Protein kinase C-beta gene variants, pathway activation, and enzastaurin activity in lung cancer
- PMID: 20439192
- PMCID: PMC3171168
- DOI: 10.3816/CLC.2010.n.021
Protein kinase C-beta gene variants, pathway activation, and enzastaurin activity in lung cancer
Abstract
Background: Protein kinase C-beta2 (PKCbeta2) is a splice-variant of the PRKCB1 gene and belongs to a family of serine/threonine-specific kinases that are predominantly activated by diacylglycerol, calcium, and phorbol ester. Cellular functions associated with PKCbeta2 activation include transformation, proliferation, and inhibition of apoptosis. Enzastaurin (LY317615) is an oral, selective, potent inhibitor of the PKCbeta2 kinase. Preclinical activity for this agent was predominantly reported in lymphoma, glioblastoma, and colorectal cancer. In patients with advanced non-small-cell lung cancer (NSCLC) whose previous therapy had failed, 13% of patients had disease control for 6 months with single-agent therapy.
Patients and methods: We investigated whether biologically relevant variants of PRKCB1 exist in lung cancer cell lines in the context of enzastaurin-induced proliferation and kinase inhibition, using exon sequencing, immunoblotting, and cytotoxicity assays in NSCLC and small-cell lung cancer (SCLC) cell lines.
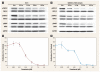
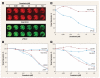
Results: We discovered a total of 6 single-nucleotide variants, but only 1 resulted in an amino acid substitution (T40I). This substitution was not located in the kinase domain of PKCbeta2 and did not affect enzastaurin's antiproliferative or phosphorylation-inhibitory activity. We found enzastaurin to be equally active in NSCLC and SCLC cell lines, with values of the 50% inhibitory concentration in a range of 0.05-0.2 microM.
Conclusion: The inhibition of phosphorylation of PKCbeta2 and the downstream molecules glycogen synthase kinase-3beta, S6RP, Akt, and forkhead transcription factor was evident in the same concentration range, which suggests the premise that the determination of phosphorylation levels of these molecules in human tissue specimens may be a useful pharmacodynamic parameter for in vivo target inhibition by enzastaurin.
Figures
Similar articles
-
The selective protein kinase C beta inhibitor enzastaurin induces apoptosis in cutaneous T-cell lymphoma cell lines through the AKT pathway.J Invest Dermatol. 2006 Jul;126(7):1641-7. doi: 10.1038/sj.jid.5700322. Epub 2006 Apr 27. J Invest Dermatol. 2006. PMID: 16645590
-
Enzastaurin (LY317615), a protein kinase Cbeta inhibitor, inhibits the AKT pathway and induces apoptosis in multiple myeloma cell lines.Mol Cancer Ther. 2006 Jul;5(7):1783-9. doi: 10.1158/1535-7163.MCT-05-0465. Mol Cancer Ther. 2006. PMID: 16891464
-
The protein kinase Cbeta-selective inhibitor, Enzastaurin (LY317615.HCl), suppresses signaling through the AKT pathway, induces apoptosis, and suppresses growth of human colon cancer and glioblastoma xenografts.Cancer Res. 2005 Aug 15;65(16):7462-9. doi: 10.1158/0008-5472.CAN-05-0071. Cancer Res. 2005. PMID: 16103100
-
Enzastaurin, a protein kinase Cbeta- selective inhibitor, and its potential application as an anticancer agent in lung cancer.Clin Cancer Res. 2007 Aug 1;13(15 Pt 2):s4641-6. doi: 10.1158/1078-0432.CCR-07-0538. Clin Cancer Res. 2007. PMID: 17671157 Review.
-
Pharmacological aspects of the enzastaurin-pemetrexed combination in non-small cell lung cancer (NSCLC).Curr Drug Targets. 2010 Jan;11(1):12-28. doi: 10.2174/138945010790031009. Curr Drug Targets. 2010. PMID: 19839931 Review.
Cited by
-
The protein kinase C (PKC) inhibitors combined with chemotherapy in the treatment of advanced non-small cell lung cancer: meta-analysis of randomized controlled trials.Clin Transl Oncol. 2015 May;17(5):371-7. doi: 10.1007/s12094-014-1241-3. Epub 2014 Oct 29. Clin Transl Oncol. 2015. PMID: 25351171 Review.
-
Chemoprevention of lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines.Chest. 2013 May;143(5 Suppl):e40S-e60S. doi: 10.1378/chest.12-2348. Chest. 2013. PMID: 23649449 Free PMC article. Review.
-
Splice variant PRKC-ζ(-PrC) is a novel biomarker of human prostate cancer.Br J Cancer. 2012 Jul 10;107(2):388-99. doi: 10.1038/bjc.2012.162. Epub 2012 May 29. Br J Cancer. 2012. PMID: 22644296 Free PMC article.
-
Discovery of prognostic biomarkers for predicting lung cancer metastasis using microarray and survival data.BMC Bioinformatics. 2015 Feb 21;16:54. doi: 10.1186/s12859-015-0463-x. BMC Bioinformatics. 2015. PMID: 25881029 Free PMC article.
-
Transcriptional analysis of apoptotic cerebellar granule neurons following rescue by gastric inhibitory polypeptide.Int J Mol Sci. 2014 Apr 1;15(4):5596-622. doi: 10.3390/ijms15045596. Int J Mol Sci. 2014. PMID: 24694544 Free PMC article.
References
-
- Griner EM, Kazanietz MG. Protein kinase C and other diacylglycerol effectors in cancer. Nat Rev Cancer. 2007;7:281–94. - PubMed
-
- Barr L, Campbell S, Baylin S. Protein kinase C-beta 2 inhibits cycling and decreases c-myc-induced apoptosis in small cell lung cancer cells. Cell Growth Differ. 1997;8:381–92. - PubMed
-
- Clark A, West K, Blumberg P, et al. Altered protein kinase C (PKC) isoforms in non-small cell lung cancer cells: PKC-delta promotes cellular survival and chemotherapeutic resistance. Cancer Res. 2003;63:780–6. - PubMed
-
- Partovian C, Simons M. Regulation of protein kinase B/Akt activity and Ser473 phosphorylation by protein kinase C-alpha in endothelial cells. Cell Signal. 2004;16:951–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

