Lack of Apobec2-related proteins causes a dystrophic muscle phenotype in zebrafish embryos
- PMID: 20440001
- PMCID: PMC2867308
- DOI: 10.1083/jcb.200912125
Lack of Apobec2-related proteins causes a dystrophic muscle phenotype in zebrafish embryos
Abstract
The chaperones Unc45b and Hsp90a are essential for folding of myosin in organisms ranging from worms to humans. We show here that zebrafish Unc45b, but not Hsp90a, binds to the putative cytidine deaminase Apobec2 (Apo2) in an interaction that requires the Unc45/Cro1p/She4p-related (UCS) and central domains of Unc45b. Morpholino oligonucleotide-mediated knockdown of the two related proteins Apo2a and Apo2b causes a dystrophic phenotype in the zebrafish skeletal musculature and impairs heart function. These phenotypic traits are shared with mutants of unc45b, but not with hsp90a mutants. Apo2a and -2b act nonredundantly and bind to each other in vitro, which suggests a heteromeric functional complex. Our results demonstrate that Unc45b and Apo2 proteins act in a Hsp90a-independent pathway that is required for integrity of the myosepta and myofiber attachment. Because the only known function of Unc45b is that of a chaperone, Apo2 proteins may be clients of Unc45b but other yet unidentified processes cannot be excluded.
Figures





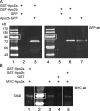
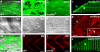

Similar articles
-
Lack of developmental redundancy between Unc45 proteins in zebrafish muscle development.PLoS One. 2012;7(11):e48861. doi: 10.1371/journal.pone.0048861. Epub 2012 Nov 7. PLoS One. 2012. PMID: 23144999 Free PMC article.
-
Shuttling of the chaperones Unc45b and Hsp90a between the A band and the Z line of the myofibril.J Cell Biol. 2008 Mar 24;180(6):1163-75. doi: 10.1083/jcb.200709128. Epub 2008 Mar 17. J Cell Biol. 2008. PMID: 18347070 Free PMC article.
-
The myosin chaperone UNC45B is involved in lens development and autosomal dominant juvenile cataract.Eur J Hum Genet. 2014 Nov;22(11):1290-7. doi: 10.1038/ejhg.2014.21. Epub 2014 Feb 19. Eur J Hum Genet. 2014. PMID: 24549050 Free PMC article.
-
UCS Chaperone Folding of the Myosin Head: A Function That Evolved before Animals and Fungi Diverged from a Common Ancestor More than a Billion Years Ago.Biomolecules. 2022 Jul 26;12(8):1028. doi: 10.3390/biom12081028. Biomolecules. 2022. PMID: 35892339 Free PMC article. Review.
-
Heat shock gene expression and function during zebrafish embryogenesis.Semin Cell Dev Biol. 2003 Oct;14(5):267-74. doi: 10.1016/j.semcdb.2003.09.018. Semin Cell Dev Biol. 2003. PMID: 14986856 Review.
Cited by
-
Elevated APOBEC3B correlates with poor outcomes for estrogen-receptor-positive breast cancers.Horm Cancer. 2014 Dec;5(6):405-13. doi: 10.1007/s12672-014-0196-8. Epub 2014 Aug 15. Horm Cancer. 2014. PMID: 25123150 Free PMC article.
-
No evidence for AID/MBD4-coupled DNA demethylation in zebrafish embryos.PLoS One. 2014 Dec 23;9(12):e114816. doi: 10.1371/journal.pone.0114816. eCollection 2014. PLoS One. 2014. PMID: 25536520 Free PMC article.
-
Engineered deaminases as a key component of DNA and RNA editing tools.Mol Ther Nucleic Acids. 2023 Oct 20;34:102062. doi: 10.1016/j.omtn.2023.102062. eCollection 2023 Dec 12. Mol Ther Nucleic Acids. 2023. PMID: 38028200 Free PMC article. Review.
-
Fam65b is important for formation of the HDAC6-dysferlin protein complex during myogenic cell differentiation.FASEB J. 2014 Jul;28(7):2955-69. doi: 10.1096/fj.13-246470. Epub 2014 Mar 31. FASEB J. 2014. PMID: 24687993 Free PMC article.
-
Mutator effects and mutation signatures of editing deaminases produced in bacteria and yeast.Biochemistry (Mosc). 2011 Jan;76(1):131-46. doi: 10.1134/s0006297911010135. Biochemistry (Mosc). 2011. PMID: 21568845 Free PMC article.
References
-
- Ai H.W., Henderson J.N., Remington S.J., Campbell R.E. 2006. Directed evolution of a monomeric, bright and photostable version of Clavularia cyan fluorescent protein: structural characterization and applications in fluorescence imaging. Biochem. J. 400:531–540 10.1042/BJ20060874 - DOI - PMC - PubMed
-
- Amali A.A., Lin C.J., Chen Y.H., Wang W.L., Gong H.Y., Rekha R.D., Lu J.K., Chen T.T., Wu J.L. 2008. Overexpression of Myostatin2 in zebrafish reduces the expression of dystrophin associated protein complex (DAPC) which leads to muscle dystrophy. J. Biomed. Sci. 15:595–604 10.1007/s11373-008-9250-2 - DOI - PubMed
-
- Anant S., Mukhopadhyay D., Sankaranand V., Kennedy S., Henderson J.O., Davidson N.O. 2001. ARCD-1, an apobec-1-related cytidine deaminase, exerts a dominant negative effect on C to U RNA editing. Am. J. Physiol. Cell Physiol. 281:C1904–C1916 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

