N-cadherin mediates plasticity-induced long-term spine stabilization
- PMID: 20440002
- PMCID: PMC2867305
- DOI: 10.1083/jcb.201003007
N-cadherin mediates plasticity-induced long-term spine stabilization
Abstract
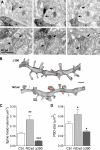
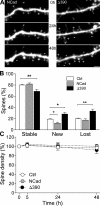
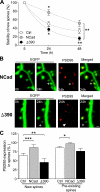
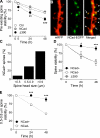
Excitatory synapses on dendritic spines are dynamic structures whose stability can vary from hours to years. However, the molecular mechanisms regulating spine persistence remain essentially unknown. In this study, we combined repetitive imaging and a gain and loss of function approach to test the role of N-cadherin (NCad) on spine stability. Expression of mutant but not wild-type NCad promotes spine turnover and formation of immature spines and interferes with the stabilization of new spines. Similarly, the long-term stability of preexisting spines is reduced when mutant NCad is expressed but enhanced in spines expressing NCad-EGFP clusters. Activity and long-term potentiation (LTP) induction selectively promote formation of NCad clusters in stimulated spines. Although activity-mediated expression of NCad-EGFP switches synapses to a highly stable state, expression of mutant NCad or short hairpin RNA-mediated knockdown of NCad prevents LTP-induced long-term stabilization of synapses. These results identify NCad as a key molecular component regulating long-term synapse persistence.
Figures


Comment in
-
N-cadherin: stabilizing synapses.J Cell Biol. 2010 May 3;189(3):397-8. doi: 10.1083/jcb.201004022. J Cell Biol. 2010. PMID: 20439992 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

