Tryptophan as a molecular shovel in the glycosyl transfer activity of Trypanosoma cruzi trans-sialidase
- PMID: 20441732
- PMCID: PMC2862183
- DOI: 10.1016/j.bpj.2010.01.006
Tryptophan as a molecular shovel in the glycosyl transfer activity of Trypanosoma cruzi trans-sialidase
Abstract
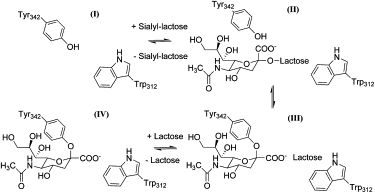
Molecular dynamics investigations into active site plasticity of Trypanosoma cruzi trans-sialidase, a protein implicated in Chagas disease, suggest that movement of the Trp(312) loop plays an important role in the enzyme's sialic acid transfer mechanism. The observed Trp(312) flexibility equates to a molecular shovel action, which leads to the expulsion of the donor aglycone leaving group from the catalytic site. These computational simulations provide detailed structural insights into sialyl transfer by the trans-sialidase and may aid the design of inhibitors effective against this neglected tropical disease.
Copyright (c) 2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Modulation of catalytic function by differential plasticity of the active site: case study of Trypanosoma cruzi trans-sialidase and Trypanosoma rangeli sialidase.Biochemistry. 2009 Apr 21;48(15):3398-406. doi: 10.1021/bi802230y. Biochemistry. 2009. PMID: 19216574 Free PMC article.
-
Preliminary 1H NMR investigation of sialic acid transfer by the trans-sialidase from Trypanosoma cruzi.Bioorg Med Chem Lett. 2000 Dec 18;10(24):2791-4. doi: 10.1016/s0960-894x(00)00572-2. Bioorg Med Chem Lett. 2000. PMID: 11133093
-
Unraveling the differences of the hydrolytic activity of Trypanosoma cruzi trans-sialidase and Trypanosoma rangeli sialidase: a quantum mechanics-molecular mechanics modeling study.J Phys Chem B. 2014 Jun 5;118(22):5807-16. doi: 10.1021/jp412294r. Epub 2014 May 21. J Phys Chem B. 2014. PMID: 24814976 Free PMC article.
-
Trypanosoma cruzi Trans-sialidase: structural features and biological implications.Subcell Biochem. 2014;74:181-201. doi: 10.1007/978-94-007-7305-9_8. Subcell Biochem. 2014. PMID: 24264246 Review.
-
Trypanosoma cruzi trans-sialidase as a drug target against Chagas disease (American trypanosomiasis).Future Med Chem. 2013 Oct;5(15):1889-900. doi: 10.4155/fmc.13.129. Future Med Chem. 2013. PMID: 24144418 Review.
Cited by
-
Evidence of ternary complex formation in Trypanosoma cruzi trans-sialidase catalysis.J Biol Chem. 2014 Jan 3;289(1):423-36. doi: 10.1074/jbc.M112.399303. Epub 2013 Nov 5. J Biol Chem. 2014. PMID: 24194520 Free PMC article.
-
Molecular Dynamics Simulations Reveal the Conformational Transition of GH33 Sialidases.Int J Mol Sci. 2023 Apr 6;24(7):6830. doi: 10.3390/ijms24076830. Int J Mol Sci. 2023. PMID: 37047800 Free PMC article.
-
Insights into the activity and specificity of Trypanosoma cruzi trans-sialidase from molecular dynamics simulations.Biochemistry. 2013 May 28;52(21):3740-51. doi: 10.1021/bi301112p. Epub 2013 May 14. Biochemistry. 2013. PMID: 23672572 Free PMC article.
-
Conformational dynamics of dry lamellar crystals of sugar based lipids: an atomistic simulation study.PLoS One. 2014 Jun 30;9(6):e101110. doi: 10.1371/journal.pone.0101110. eCollection 2014. PLoS One. 2014. PMID: 24978205 Free PMC article.
-
Analysis of Glycan Recognition by Concanavalin A Using Absolute Binding Free Energy Calculations.J Chem Inf Model. 2024 Oct 28;64(20):8063-8073. doi: 10.1021/acs.jcim.4c01088. Epub 2024 Oct 16. J Chem Inf Model. 2024. PMID: 39413277 Free PMC article.
References
-
- Montfort W.R., Perry K.M., Stroud R.M. Structure, multiple site binding, and segmental accommodation in thymidylate synthase on binding dUMP and an anti-folate. Biochemistry. 1990;29:6964–6977. - PubMed
-
- Rose R.B., Craik C.S., Stroud R.M. Domain flexibility in retroviral proteases: structural implications for drug resistant mutations. Biochemistry. 1998;37:2607–2621. - PubMed
-
- Amaro R.E., Minh D.D.L., McCammon J.A. Remarkable loop flexibility in avian influenza N1 and its implications for antiviral drug design. J. Am. Chem. Soc. 2007;129:7764–7765. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

