Accumulation of oxidized proteins in Herpesvirus infected cells
- PMID: 20441790
- PMCID: PMC3206308
- DOI: 10.1016/j.freeradbiomed.2010.04.026
Accumulation of oxidized proteins in Herpesvirus infected cells
Abstract
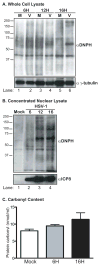
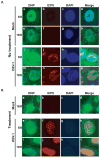
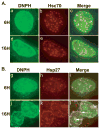
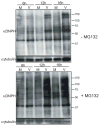
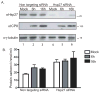
Oxidative stress gives rise to an environment that can be highly damaging to proteins, lipids, and DNA. Previous studies indicate that Herpesvirus infections cause oxidative stress in cells and in tissues. The biological consequences of virus-induced oxidative stress have not been characterized. Studies from many groups indicate that proteins which have been damaged through oxidative imbalances are either degraded by the 20S proteasome in a ubiquitin-independent fashion or form aggregates that are resistant to proteolysis. We have previously shown that herpes simplex virus type 1 (HSV-1) replication was significantly enhanced in the presence of the cellular antioxidant chaperone Hsp27, indicating a possible role for this protein in managing virus-induced oxidative stress. Here we show that oxidized proteins accumulate during infections with two distantly related herpesviruses, HSV-1 and Rhesus Rhadinovirus (RRV), a close relative of the Kaposi's sarcoma-associated herpesvirus (KSHV). The presence of oxidized proteins was not entirely unexpected as oxidative stress during herpesvirus infection has been previously documented. Unexpectedly, some oxidized proteins are removed in a proteasome-dependent fashion throughout infection and others resist degradation. Oxidized proteins that resist proteolysis become sequestered in foci within the nucleus and are not associated with virus-induced chaperone enriched domains (VICE), active centers of protein quality control, but rather coincide with Hsp27-enriched foci that were previously described by our laboratory. Experiments also indicate that the accumulation of oxidized proteins is more pronounced in cells depleted for Hsp27. We propose that Hsp27 may facilitate oxidized protein turnover at VICE domains in the nucleus during infection. Hsp27 may also buffer toxic effects of highly-carbonylated, defective proteins that resist proteolysis by promoting their aggregation in the nucleus. These roles of Hsp27 during virus infection are most likely not mutually exclusive.
Copyright 2010 Elsevier Inc. All rights reserved.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

