The polarized distribution of Na+,K+-ATPase: role of the interaction between {beta} subunits
- PMID: 20444976
- PMCID: PMC2893986
- DOI: 10.1091/mbc.e10-01-0081
The polarized distribution of Na+,K+-ATPase: role of the interaction between {beta} subunits
Abstract
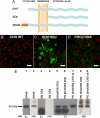
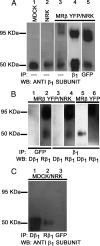
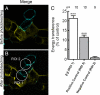
The very existence of higher metazoans depends on the vectorial transport of substances across epithelia. A crucial element of this transport is the membrane enzyme Na(+),K(+)-ATPase. Not only is this enzyme distributed in a polarized manner in a restricted domain of the plasma membrane but also it creates the ionic gradients that drive the net movement of glucose, amino acids, and ions across the entire epithelium. In a previous work, we have shown that Na(+),K(+)-ATPase polarity depends on interactions between the beta subunits of Na(+),K(+)-ATPases located on neighboring cells and that these interactions anchor the entire enzyme at the borders of the intercellular space. In the present study, we used fluorescence resonance energy transfer and coprecipitation methods to demonstrate that these beta subunits have sufficient proximity and affinity to permit a direct interaction, without requiring any additional extracellular molecules to span the distance.
Figures



Similar articles
-
beta-Subunit overexpression alters the stoicheometry of assembled Na-K-ATPase subunits in MDCK cells.Am J Physiol Renal Physiol. 2008 Nov;295(5):F1314-23. doi: 10.1152/ajprenal.90406.2008. Epub 2008 Aug 13. Am J Physiol Renal Physiol. 2008. PMID: 18701620 Free PMC article.
-
The O-glycosylated ectodomain of FXYD5 impairs adhesion by disrupting cell-cell trans-dimerization of Na,K-ATPase β1 subunits.J Cell Sci. 2016 Jun 15;129(12):2394-406. doi: 10.1242/jcs.186148. Epub 2016 May 3. J Cell Sci. 2016. PMID: 27142834 Free PMC article.
-
The Na+-K+-ATPase as self-adhesion molecule and hormone receptor.Am J Physiol Cell Physiol. 2012 Feb 1;302(3):C473-81. doi: 10.1152/ajpcell.00083.2011. Epub 2011 Nov 2. Am J Physiol Cell Physiol. 2012. PMID: 22049208 Review.
-
The polarized expression of Na+,K+-ATPase in epithelia depends on the association between beta-subunits located in neighboring cells.Mol Biol Cell. 2005 Mar;16(3):1071-81. doi: 10.1091/mbc.e04-03-0267. Epub 2004 Dec 22. Mol Biol Cell. 2005. PMID: 15616198 Free PMC article.
-
The Na-K-ATPase α₁β₁ heterodimer as a cell adhesion molecule in epithelia.Am J Physiol Cell Physiol. 2012 May 1;302(9):C1271-81. doi: 10.1152/ajpcell.00456.2011. Epub 2012 Jan 25. Am J Physiol Cell Physiol. 2012. PMID: 22277755 Free PMC article. Review.
Cited by
-
β1-Na(+),K(+)-ATPase gene therapy upregulates tight junctions to rescue lipopolysaccharide-induced acute lung injury.Gene Ther. 2016 Jun;23(6):489-99. doi: 10.1038/gt.2016.19. Epub 2016 Mar 17. Gene Ther. 2016. PMID: 26910760
-
The mitochondrial Cu+ transporter PiC2 (SLC25A3) is a target of MTF1 and contributes to the development of skeletal muscle in vitro.Front Mol Biosci. 2022 Nov 9;9:1037941. doi: 10.3389/fmolb.2022.1037941. eCollection 2022. Front Mol Biosci. 2022. PMID: 36438658 Free PMC article.
-
Quantitative Proteomics of Clinically Relevant Drug-Metabolizing Enzymes and Drug Transporters and Their Intercorrelations in the Human Small Intestine.Drug Metab Dispos. 2020 Apr;48(4):245-254. doi: 10.1124/dmd.119.089656. Epub 2020 Jan 20. Drug Metab Dispos. 2020. PMID: 31959703 Free PMC article.
-
Cardiac Glycosides: From Natural Defense Molecules to Emerging Therapeutic Agents.Biomolecules. 2025 Jun 17;15(6):885. doi: 10.3390/biom15060885. Biomolecules. 2025. PMID: 40563525 Free PMC article. Review.
-
21-Benzylidene digoxin: a proapoptotic cardenolide of cancer cells that up-regulates Na,K-ATPase and epithelial tight junctions.PLoS One. 2014 Oct 7;9(10):e108776. doi: 10.1371/journal.pone.0108776. eCollection 2014. PLoS One. 2014. PMID: 25290152 Free PMC article.
References
-
- Arrate M. P., Rodriguez J. M., Tran T. M., Brock T. A., Cunningham S. A. Cloning of human junctional adhesion molecule 3 (JAM3) and its identification as the JAM2 counter-receptor. J. Biol. Chem. 2001;276:45826–45832. - PubMed
-
- Bab-Dinitz E., Albeck S., Peleg Y., Brumfeld J., Gottschalk K. E., Karlish S. A C-terminal lobe of the β subunit of Na, K-ATPase and H, K-ATPase resembles cell adhesion molecules. Biochemistry. 2009;48:8684–8691. - PubMed
-
- Belusa R., Wang Z., Matsubara T., Sahingren B., Dulubova I., Nair A. C., Ruoslathi E., Greengard P., Aperia A. “Mutation of the protein kinase C phosphorylation site on rat α1 Na+, K+-ATPase alters regulation of intracellular Na+ and pH and influences Cell shape and adhesiveness”. J. Biol. Chem. 1997;272:20179–20184. - PubMed
-
- Caplan M. J., Anderson H. C., Palade G. E., Jamieson J. D. Intracellular sorting and polarized cell surface delivery of (Na+, K+)ATPase, an endogenous component of MDCK cell basolateral plasma membranes. Cell. 1986;46:623–631. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

