Changes in S1 neural responses during tactile discrimination learning
- PMID: 20445033
- PMCID: PMC2904220
- DOI: 10.1152/jn.00194.2010
Changes in S1 neural responses during tactile discrimination learning
Abstract
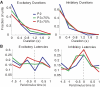
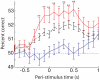
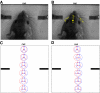
In freely moving rats that are actively performing a discrimination task, single-unit responses in primary somatosensory cortex (S1) are strikingly different from responses to comparable tactile stimuli in immobile rats. For example, in the active discrimination context prestimulus response modulations are common, responses are longer in duration and more likely to be inhibited. To determine whether these differences emerge as rats learned a whisker-dependent discrimination task, we recorded single-unit S1 activity while rats learned to discriminate aperture-widths using their whiskers. Even before discrimination training began, S1 responses in freely moving rats showed many of the signatures of active responses, such as increased duration of response and prestimulus response modulations. As rats subsequently learned the discrimination task, single unit responses changed: more cortical units responded to the stimuli, neuronal sensory responses grew in duration, and individual neurons better predicted aperture-width. In summary, the operant behavioral context changes S1 tactile responses even in the absence of tactile discrimination, whereas subsequent width discrimination learning refines the S1 representation of aperture-width.
Figures




Similar articles
-
Behavioral properties of the trigeminal somatosensory system in rats performing whisker-dependent tactile discriminations.J Neurosci. 2001 Aug 1;21(15):5752-63. doi: 10.1523/JNEUROSCI.21-15-05752.2001. J Neurosci. 2001. PMID: 11466447 Free PMC article.
-
Layer-specific somatosensory cortical activation during active tactile discrimination.Science. 2004 Jun 25;304(5679):1989-92. doi: 10.1126/science.1093318. Science. 2004. PMID: 15218154
-
Bilateral Discrimination of Tactile Patterns without Whisking in Freely Running Rats.J Neurosci. 2017 Aug 9;37(32):7567-7579. doi: 10.1523/JNEUROSCI.0528-17.2017. Epub 2017 Jun 29. J Neurosci. 2017. PMID: 28663200 Free PMC article.
-
Multielectrode Recordings in the Somatosensory System.In: Nicolelis MAL, editor. Methods for Neural Ensemble Recordings. 2nd edition. Boca Raton (FL): CRC Press/Taylor & Francis; 2008. Chapter 6. In: Nicolelis MAL, editor. Methods for Neural Ensemble Recordings. 2nd edition. Boca Raton (FL): CRC Press/Taylor & Francis; 2008. Chapter 6. PMID: 21204443 Free Books & Documents. Review.
-
Neocortical dynamics during whisker-based sensory discrimination in head-restrained mice.Neuroscience. 2018 Jan 1;368:57-69. doi: 10.1016/j.neuroscience.2017.09.003. Epub 2017 Sep 14. Neuroscience. 2018. PMID: 28919043 Free PMC article. Review.
Cited by
-
Frequency-specific coupling in fronto-parieto-occipital cortical circuits underlie active tactile discrimination.Sci Rep. 2019 Mar 25;9(1):5105. doi: 10.1038/s41598-019-41516-3. Sci Rep. 2019. PMID: 30911025 Free PMC article.
-
A Brain to Spine Interface for Transferring Artificial Sensory Information.Sci Rep. 2020 Jan 21;10(1):900. doi: 10.1038/s41598-020-57617-3. Sci Rep. 2020. PMID: 31964948 Free PMC article.
-
Distinct learning-induced changes in stimulus selectivity and interactions of GABAergic interneuron classes in visual cortex.Nat Neurosci. 2018 Jun;21(6):851-859. doi: 10.1038/s41593-018-0143-z. Epub 2018 May 21. Nat Neurosci. 2018. PMID: 29786081 Free PMC article.
-
Behaviour-dependent recruitment of long-range projection neurons in somatosensory cortex.Nature. 2013 Jul 18;499(7458):336-40. doi: 10.1038/nature12236. Epub 2013 Jun 23. Nature. 2013. PMID: 23792559
-
Learning and attention increase visual response selectivity through distinct mechanisms.Neuron. 2022 Feb 16;110(4):686-697.e6. doi: 10.1016/j.neuron.2021.11.016. Epub 2021 Dec 13. Neuron. 2022. PMID: 34906356 Free PMC article.
References
-
- Abdi H. (Editor). Bonferroni and Šidák Corrections for Multiple Comparisons. Thousand Oaks, CA: Sage, 2007
-
- Bao S, Chang EF, Woods J, Merzenich MM. Temporal plasticity in the primary auditory cortex induced by operant perceptual learning. Nat Neurosci 7: 974–981, 2004 - PubMed
-
- Butovas S, Schwarz C. Detection psychophysics of intracortical microstimulation in rat primary somatosensory cortex. Eur J Neurosci 25: 2161–2169, 2007 - PubMed
-
- Butt AE, Chavez CM, Flesher MM, Kinney-Hurd BL, Araujo GC, Miasnikov AA, Weinberger NM. Association learning-dependent increases in acetylcholine release in the rat auditory cortex during auditory classical conditioning. Neurobiol Learn Mem 92: 400–409, 2009 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

