Phymatotrichum (cotton) root rot caused by Phymatotrichopsis omnivora: retrospects and prospects
- PMID: 20447281
- PMCID: PMC6640249
- DOI: 10.1111/j.1364-3703.2010.00616.x
Phymatotrichum (cotton) root rot caused by Phymatotrichopsis omnivora: retrospects and prospects
Abstract
Phymatotrichum (cotton or Texas) root rot is caused by the soil-borne fungus Phymatotrichopsis omnivora (Duggar) Hennebert. The broad host range of the fungus includes numerous crop plants, such as alfalfa and cotton. Together with an overview of existing knowledge, this review is aimed at discussing the recent molecular and genomic approaches that have been undertaken to better understand the disease development at the molecular level with the ultimate goal of developing resistant germplasm.
Taxonomy: Phymatotrichopsis omnivora (Duggar) Hennebert [synonym Phymatotrichum omnivorum (Shear) Duggar] is an asexual fungus with no known sexual stage. Mitosporic botryoblastospores occasionally form on epigeous spore mats in nature, but perform no known function and do not contribute to the disease cycle. The fungus has been affiliated erroneously with the polypore basidiomycete Sistotrema brinkmannii (Bres.) J. Erikss. Recent phylogenetic studies have placed this fungus in the ascomycete order Pezizales. HOST RANGE AND DISEASE SYMPTOMS: The fungus infects most dicotyledonous field crops, causing significant losses to cotton, alfalfa, grape, fruit and nut trees and ornamental shrubs in the south-western USA, northern Mexico and possibly parts of central Asia. However, this fungus does not cause disease in monocotyledonous plants. Symptoms include an expanding tissue collapse (rot) of infected taproots. In above-ground tissues, the root rot results in vascular discoloration of the stem and rapid wilting of the leaves without abscission, and eventually the death of the plant. Characteristic mycelial strands of the pathogen are typically present on the root's surface, aiding diagnosis.
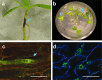
Pathogenicity: Confocal imaging of P. omnivora interactions with Medicago truncatula roots revealed that infecting hyphae do not form any specialized structures for penetration and mainly colonize cortical cells and eventually form a mycelial mantle covering the root's surfaces. Cell wall-degrading enzymes have been implicated in penetration and symptom development. Global gene expression profiling of infected M. truncatula revealed roles for jasmonic acid, ethylene and the flavonoid pathway during disease development. Phymatotrichopsis omnivora apparently evades induced host defences and may suppress the host's phytochemical defences at later stages of infection to favour pathogenesis.
Disease control: No consistently effective control measures are known. The long-lived sclerotia and facultative saprotrophism of P. omnivora make crop rotation ineffective. Chemical fumigation methods are not cost-effective for most crops. Interestingly, no genetic resistance has been reported in any of the susceptible crop species.
Figures


Similar articles
-
Global gene expression profiling during Medicago truncatula-Phymatotrichopsis omnivora interaction reveals a role for jasmonic acid, ethylene, and the flavonoid pathway in disease development.Mol Plant Microbe Interact. 2009 Jan;22(1):7-17. doi: 10.1094/MPMI-22-1-0007. Mol Plant Microbe Interact. 2009. PMID: 19061398
-
Molecular systematics of the cotton root rot pathogen, Phymatotrichopsis omnivora.Persoonia. 2009 Jun;22:63-74. doi: 10.3767/003158509X430930. Epub 2009 Mar 11. Persoonia. 2009. PMID: 20198139 Free PMC article.
-
The First Genomic Resources for Phymatotrichopsis omnivora, a Soilborne Pezizomycete Pathogen with a Broad Host Range.Phytopathology. 2021 Oct;111(10):1897-1900. doi: 10.1094/PHYTO-01-21-0014-A. Epub 2021 Nov 8. Phytopathology. 2021. PMID: 33728936
-
Developing tools to unravel the biological secrets of Rosellinia necatrix, an emergent threat to woody crops.Mol Plant Pathol. 2012 Apr;13(3):226-39. doi: 10.1111/j.1364-3703.2011.00753.x. Epub 2011 Oct 20. Mol Plant Pathol. 2012. PMID: 22014332 Free PMC article. Review.
-
Tobacco leaf spot and root rot caused by Rhizoctonia solani Kühn.Mol Plant Pathol. 2011 Apr;12(3):209-16. doi: 10.1111/j.1364-3703.2010.00664.x. Epub 2010 Oct 1. Mol Plant Pathol. 2011. PMID: 21355993 Free PMC article. Review.
Cited by
-
The Pattern Recognition Receptor FLS2 Can Shape the Arabidopsis Rhizosphere Microbiome β-Diversity but Not EFR1 and CERK1.Plants (Basel). 2022 May 17;11(10):1323. doi: 10.3390/plants11101323. Plants (Basel). 2022. PMID: 35631748 Free PMC article.
-
Recent Advances in Microfluidics for the Early Detection of Plant Diseases in Vegetables, Fruits, and Grains Caused by Bacteria, Fungi, and Viruses.J Agric Food Chem. 2024 Jul 17;72(28):15401-15415. doi: 10.1021/acs.jafc.4c00454. Epub 2024 Jun 14. J Agric Food Chem. 2024. PMID: 38875493 Free PMC article. Review.
-
Development of a rapid, sensitive, and field-deployable razor ex BioDetection system and quantitative PCR assay for detection of Phymatotrichopsis omnivora using multiple gene targets.Appl Environ Microbiol. 2013 Apr;79(7):2312-20. doi: 10.1128/AEM.03239-12. Epub 2013 Jan 25. Appl Environ Microbiol. 2013. PMID: 23354717 Free PMC article.
-
Genomics of Plant Disease Resistance in Legumes.Front Plant Sci. 2019 Oct 30;10:1345. doi: 10.3389/fpls.2019.01345. eCollection 2019. Front Plant Sci. 2019. PMID: 31749817 Free PMC article. Review.
-
Pest categorisation of Phymatotrichopsis omnivora.EFSA J. 2019 Mar 12;17(3):e05619. doi: 10.2903/j.efsa.2019.5619. eCollection 2019 Mar. EFSA J. 2019. PMID: 32626246 Free PMC article.
References
-
- Alderman, S.C. and Hine, R.B. (1982) Vertical distribution in soil and induction of disease by strands of Phymatotrichum omnivorum . Phytopathology, 72, 409–412.
-
- Alderman, S.C. and Stowell, L.J. (1986) Strand ontogeny in Phymatotrichum omnivorum . Trans. Br. Mycol. Soc. 86, 207–211.
-
- Baniecki, J.F. and Bloss, H.E. (1969) The basidial stage of Phymatotrichum omnivorum . Mycologia, 61, 1054–1059. - PubMed
-
- Bird, L.S. , Bush, D.L. , Percy, R.G. , Bourland, F.M. and Hood, J.E. (1977) Resistance to Phymatotrichum root rot in cotton. Proc. Beltwide Cotton Prod. Res. Conf. 37, 229–232.
-
- Bird, L.S. , El‐Zik, K.M. , Thaxton, P.M. , Percy, R.G. and Lazo, G.R. (1980) Status of resistance to Phymatotrichum root rot in multi‐adversity resistance cottons. Proc. Beltwide Cotton Prod. Res. Conf. 40, 277–279.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

